 |
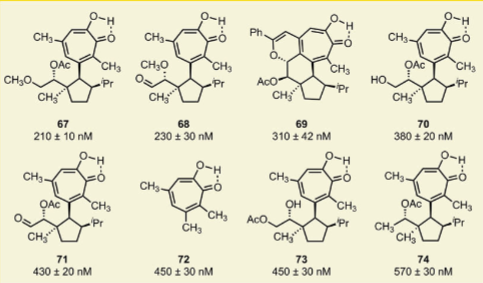
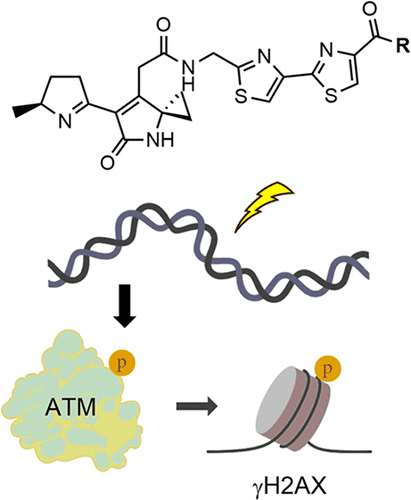
Airan Y, Fedorova O, Heer C, Rees MG, Ronan MM, Roth JA, Bindra RS, Herzon SB. (2025) Synthesis and biological evaluation of colibactin derivatives. ACS Chem Biol, 21(1):26-36, 19 Dec 2025. PMID: 41419211 | PMCID: PMC12856499 |
 |
Gupta V, Wang Z, Combs JB, Wright T, Chen L, Lin B, Holmes R, Qin B, Oh J, Crawford JM, Herzon SB. (2025) Three-component assembly and structure-function relationships of (-)-gukulenin A. Science, 390(6778):eaea9310, 11 Dec 2025. PMID: 41231967 | PMCID: PMC12863052
|
 |
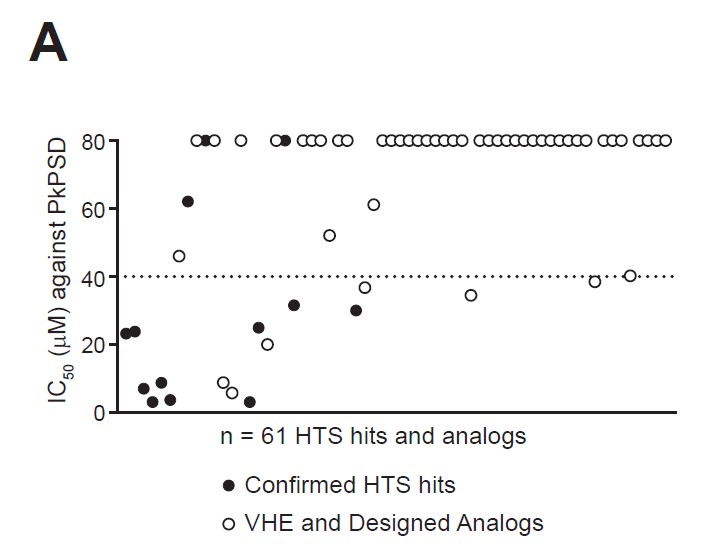
Long K, Bhattacharjee D, Newman-Stonebraker SH, Suhr S, Mercado BQ, Tighe A, Romero L, Thompson SL, Sausville EL, John KM, Julian L, Mishra S, Klingbeil O, Gupta P, Bhatt U, Gao AC, Ricardo S, Vakoc CR, Bornhauser BC, […] Sheltzer JM. (2025) A clinical-stage oncology compound selectively targets drug-resistant cancers. bioRxiv, 30 Nov 2025. PPR: PPR1128338
|
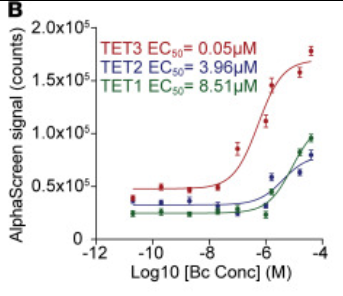 |
Liu B, Dai Y, Wang Z, Song J, Du Y, Lv H, Bellone S, Yang YH, Kennedy A, Zhang S, Venkatachalapathy M, Surovtseva Y, Wang P, Carmichael GG, Taylor HS, Zhang X, Li D, Huang Y. (2025) TET3 is a common epigenetic immunomodulator of pathogenic macrophages. J Clin Invest, 135(21):e194879, 12 Aug 2025. PMID: 40794443 | PMCID: PMC12578387
|
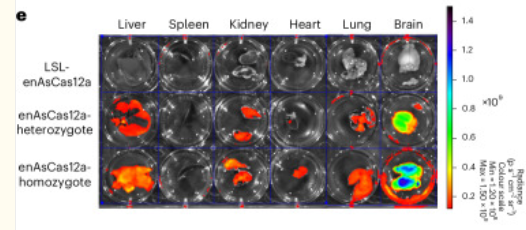 |
Tang K, Zhou L, Tian X, Fang SY, Vandenbulcke E, Du A, Shen J, Cao H, Zhou J, Chen K, Kim HR, Luo Z, Xin S, Lin SH, Park D, Yang L, Zhang Y, Suzuki K, Majety M, […] Chen S. (2025) Cas12a-knock-in mice for multiplexed genome editing, disease modelling and immune-cell engineering. Nat Biomed Eng, 9(8):1290-1308, 20 Mar 2025. PMID: 40114032 | PMCID: PMC12360953
|
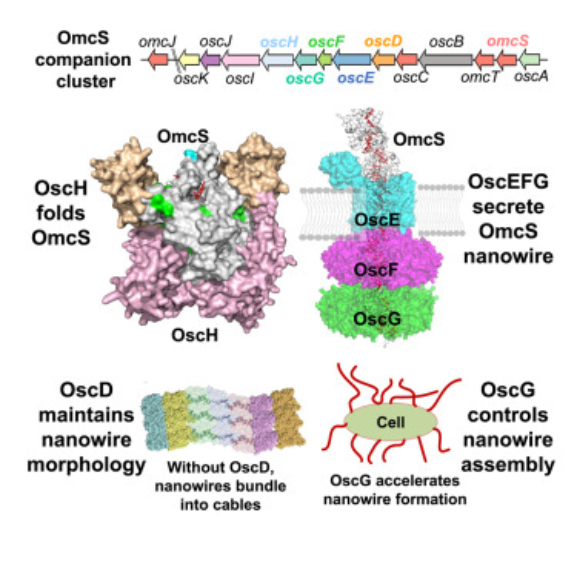 |
Shen C, Salazar-Morales AI, Jung W, Erwin J, Gu Y, Coelho A, Gupta K, Yalcin SE, Samatey FA, Malvankar NS. (2025) A widespread and ancient bacterial machinery assembles cytochrome OmcS nanowires essential for extracellular electron transfer. Cell Chem Biol, 32(2):239-254.e7, 15 Jan 2025.
|
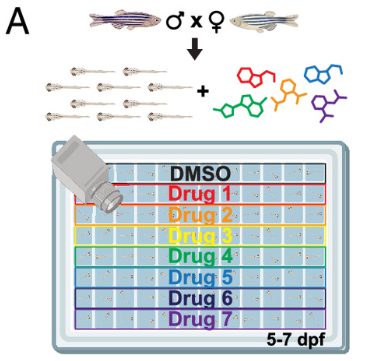 |
Jamadagni P, Dai Y, Liu Y, Weinschutz Mendes H, Pruitt A, Khan S, Yang L, Huang TC, Huang X, Deans PJM, Balafkan N, Zhao D, Xu G, Liu Y, Li N, Wu W, Fitzpatrick SE, Neelakantan U, Chen T, Szialta C, Jin DS, Lacadie CM, Umlauf S, Papademetris X, Surovtseva YV, Brennand KJ, Wang Z, Hoffman EJ. Pharmaco-behavioral profiling identifies suppressors of autism gene-associated phenotypes in zebrafish. Proc Natl Acad Sci U S A. 2026 Mar 24;123(12):e2518846123. doi: 10.1073/pnas.2518846123. Epub 2026 Mar 16. PMID: 41838920
|
 |
Liu B, Dai Y, Wang Z, Song J, Du Y, Lv H, Bellone S, Yang YH, Kennedy A, Zhang S, Venkatachalapathy M, Surovtseva YV, Wang P, Carmichael GG, Taylor HS, Zhang X, Li D, Huang Y. TET3 is a common epigenetic immunomodulator of pathogenic macrophages. J Clin Invest. 2025 Aug 12:e194879. PMID: 40794443 |
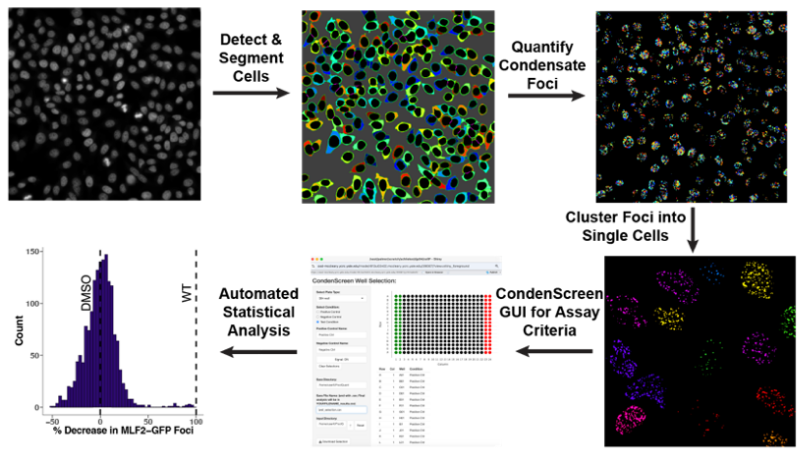 |
Poch D, Mukherjee C, Mallik S, Todorow V, Kuiper EFE, Dhingra N, Surovtseva YV, Schlieker C. Integrative chemical genetics platform identifies condensate modulators linked to neurological disorders. bioRxiv [Preprint]. 2025 Jun 8:2025.06.07.658469. PMID: 40502095 PMCID: PMC12157464
|
 |
Liu T, Xu L, Chung K, Sisto LJ, Hwang J, Zhang C, Van Zandt MC, Pyle AM. Molecular insights into de novo small-molecule recognition by an intron RNA structure. Proc Natl Acad Sci USA. 2025 May 13;122(19):e2502425122. PMID: 40339124 PMCID: PMC12088405
|
 |
Ranjan K, Rajendran BK, Deen IU, Costantini A, de Rodas ML, Desai SS, Scallo F, Gianino N, Ferrone S, Schalper KA. IL-4 mediated TAP2 downregulation is a dominant and reversible mechanism of immune evasion and immunotherapy resistance in non-small cell lung cancer. Mol Cancer. 2025 Mar 17;24(1):80. PMID: 40091029 PMCID: PMC11912681
|
 |
Brown TJ, Pichurin J, Parrado CR, Kabeche L, Baserga SJ. A role for the kinetochore protein, NUF2, in ribosome biogenesis. Mol Biol Cell. 2025 Feb 1;36(2):ar16. PMID: 39705402 PMCID: PMC11809303
|
 |
Arias Escayola D, Zhang C, Nischwitz E, Schärfen L, Dörner K, Straube K, Kutay U, Butter F, Neugebauer KM. Identification of coilin interactors reveals coordinated control of Cajal body number and structure. J Cell Biol. 2025 Feb 3;224(2):e202305081. PMID: 39602297 PMCID: PMC11602656
|
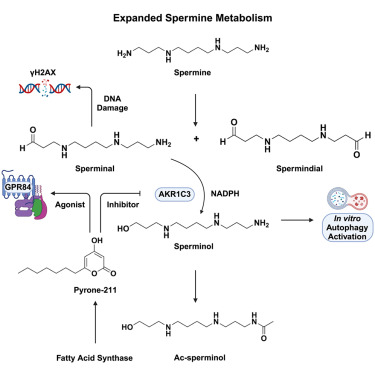 |
Dudkina N, Park HB, Song D, Jain A, Khan SA, Flavell RA, Johnson CH, Palm NW, Crawford JM. Human AKR1C3 binds agonists of GPR84 and participates in an expanded polyamine pathway. Cell Chem Biol 2024. PMID: 39163853 PMCID: PMC11748234
|
 |
Gunasekharan V, Lin HK, Marczyk M, Rios-Hoyo A, Campos GE, Shan NL, Ahmed M, Umlauf S, Gareiss P, Raaisa R, Williams R, Cardone R, Siebel S, Kibbey R, Surovtseva YV, Pusztai L. Phosphoenolpyruvate carboxykinase-2 (PCK2) is a therapeutic target in triple-negative breast cancer. Breast Cancer Res Treat 2024. PMID: 39177932
|
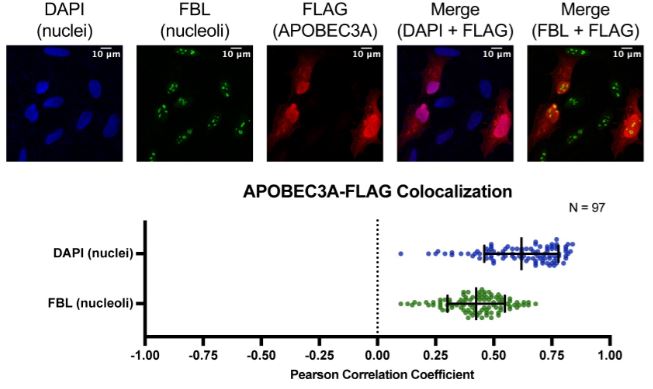 |
McCool MA, Bryant CJ, Abriola L, Surovtseva YV, Baserga SJ. The cytidine deaminase APOBEC3A regulates nucleolar function to promote cell growth and ribosome biogenesis. PLoS Biol 2024. PMID: 38976757; PMCID: PMC11257408
|
 |
Li J, Leng L, Pantouris G, Manjula R, Piecychna M, Abriola L, Hu B, Lolis E, Armstrong ME, Donnelly SC, Bucala R. A small-molecule allele-selective transcriptional inhibitor of the MIF immune susceptibility locus. J Biol Chem 2024. PMID: 38838773; PMCID: PMC11259703
|
 |
Dousa KM, Shin E, Kurz SG, Plummer M, Nantongo M, Bethel CR, Taracila MA, Nguyen DC, Kreiswith BN, Daley CL,Remy KE, Holland SM, Bonomo RA. Synergistic effects of sulopenem in combination with cefuroxime or durlobactam against Mycobacterium abscessus. mBio 2024. PMID: 38742824; PMCID: PMC11237399
|
 |
Gomes JV, Singh-Bhagania S, Cenci M, Chacon Cordon C, Singh M, Butterwick JA. The molecular basis of sugar detection by an insect taste receptor. Nature 2024. PMID: 38447670; PMCID: PMC11062906
|
 |
Bryant CJ, McCool MA, Rosado González GT, Abriola L, Surovtseva YV, Baserga SJ. Discovery of novel microRNA mimic repressors of ribosome biogenesis. Nucleic Acids Res 2024. PMID: 38197221; PMCID: PMC10899765
|
 |
Vydyam P, Choi JY, Gihaz S, Chand M, Gewirtz M, Thekkiniath J, Lonardi S, Gennaro JC, Ben Mamoun C. Babesia BdFE1esterase is required for the anti-parasitic activity of the ACE inhibitor fosinopril. J Biol Chem 2023. PMID: 37797695; PMCID: PMC10663679
|
 |
Sokolov AM, Aurich M, Bordey A. In utero electroporated neurons for medium-throughput screening of compounds regulating neuron morphology. eNeuro 2023. PMID: 37620147; PMCID: PMC10464655
|
 |
Wei J, Alfajaro MM, Cai WL, Graziano VR, Strine MS, Filler RB, Biering SB, Sarnik SA, Patel S, Menasche BL, Compton SR, Konermann S, Hsu PD, Orchard RC, Yan Q, Wilen CB. The KDM6A-KMT2D-p300 axis regulates susceptibility to diverse coronaviruses by mediating viral receptor expression. PLoS Pathog 2023. PMID: 37410700; PMCID: PMC10325096
|
 |
Wang BZ, Nash TR, Zhang X, Rao J, Abriola L, Kim Y, Zakharov S, Kim M, Luo LJ, Morsink M, Liu B, Lock RI, Fleisher S, Tamargo MA, Bohnen M, Welch CL, Chung WK, Marx SO, Surovtseva YV, Vunjak-Novakovic G, Fine BM. Engineered cardiac tissue model of restrictive cardiomyopathy for drug discovery. Cell Rep Med. 2023 Mar 21;4(3):100976. doi: 10.1016/j.xcrm.2023.100976. Epub 2023 Mar 14.
|
 |
Bryant CJ, McCool MA, Rosado-Gonzalez GT, Abriola L, Surovtseva YV, Baserga SJ. Discovery of novel microRNA regulators of ribosome biogenesis. bioRxiv. 2023 Feb 17;2023.02.17.526327. doi: 10.1101/2023.02.17.526327. |
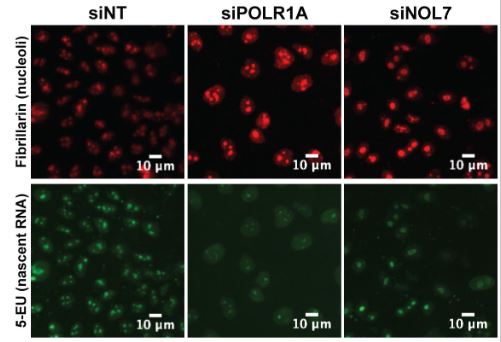 |
McCool MA, Bryant CJ, Huang H, Ogawa LM, Farley-Barnes KI, Sondalle SB, Abriola L, Surovstseva YV, Baserga SJ. Human nucleolar protein 7 (NOL7) is required for early pre-rRNA accumulation and pre-18S rRNA processing. RNA Biol. 2023 Jan; 20(1):257-271. doi: 10.1080/15476286.2023.2217392.
|
 |
Shillingford S, Zhang L, Surovtseva Y, Dorry S, Lolis E, Bennett AM. A novel site on dual-specificity phosphatase MKP7/DUSP16 is required for catalysis and MAPK binding. J Biol Chem. 2022 Dec;298(12):102617. doi: 10.1016/j.jbc.2022.102617. Epub 2022 Oct 19.
|
 |
McCool MA, Buhagiar AF, Bryant CJ, Ogawa LM, Abriola L, Surovstseva YV, Baserga SJ. Human pre-60S assembly factors link rRNA transcription to pre-rRNA processing. RNA. 2022 Nov 2;29(1):82-96. doi: 10.1261/rna.079149.122.
|
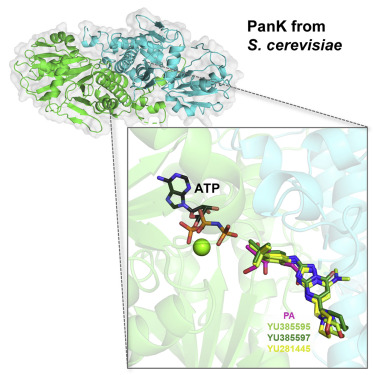 |
Gihaz S, Gareiss P, Choi J-Y, Renard I, Pal AC, Surovtseva Y, Chiu JE, Thekkiniath J, Plummer M, Hungerford W, Montgomery ML, Hosford A, Adams EM, Lightfoot JD, Fox DF, Ojo KK, Staker BL, Fuller K, Mamoun CB. High-resolution crystal structure and chemical screening reveal panthothenate kinase as a new target for antifungal treatment. Structure. 2022 Sep 21;S0969-2126(22)00354-9. doi: 10.1016/j.str.2022.09.001. PMID: 36167065
|
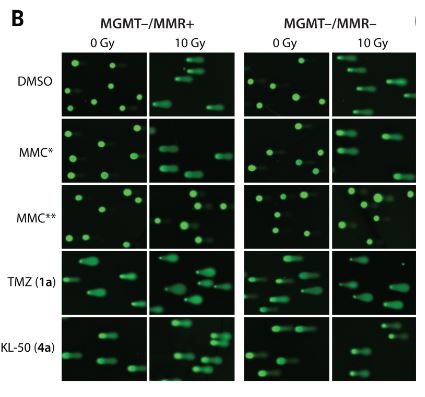 |
Lin K, Gueble SE, Sundaram RK, Huseman ED, Bindra RS, Herzon SB. Mechanism-based design of agents that selectively target drug-resistant glioma. Science . 2022 Jul 29;377(6605):502-511. doi: 10.1126/science.abn7570. Epub 2022 Jul 28. PMID: 35901163
|
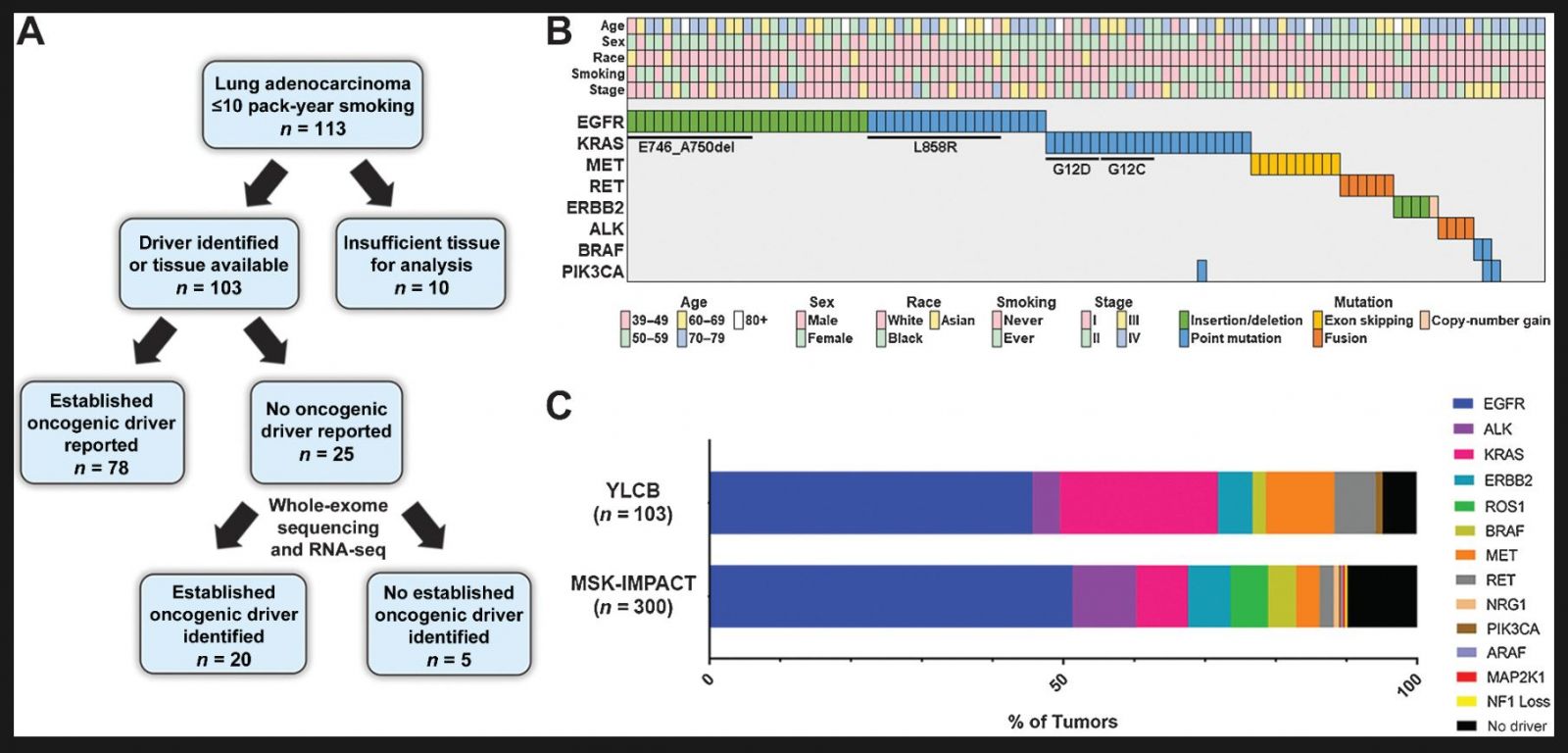 |
Hunihan L, Zhao D, Lazowski H, Li M, Qian Y, Abriola L, Surovtseva YV, Muthusamy V, Tanoue LT, Rothberg BEG, Schalper KA, Herbst RS, Wilson FH. RASGRF1 Fusions Activate Oncogenic RAS Signaling and Confer Sensitivity to MEK Inhibition. Clin Cancer Res. 2022 Jul 15;28(14):3091-3103. doi: 10.1158/1078-0432.CCR-21-4291. PMID: 35247929
|
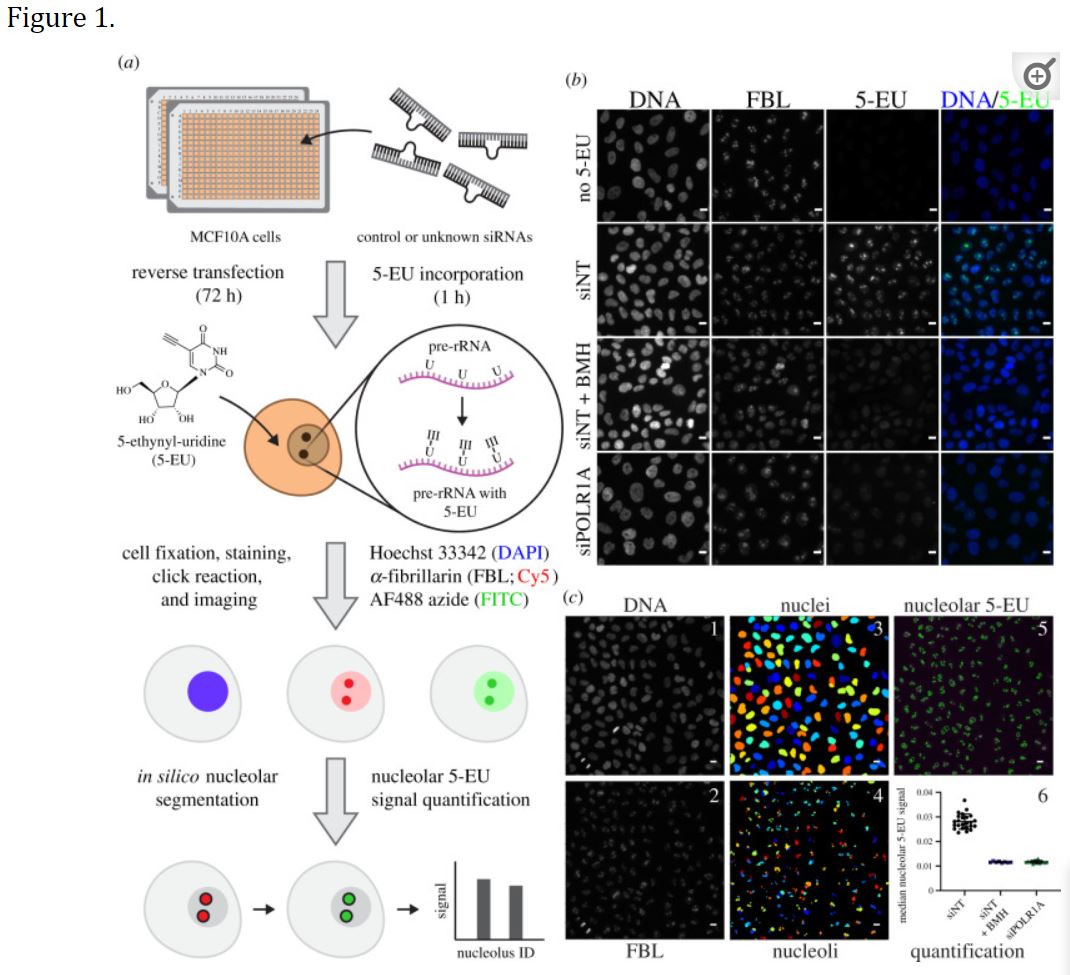 |
Bryant CJ, McCool MA, Abriola L, Surovtseva YV, Baserga SJ. A high-throughput assay for directly monitoring nucleolar rRNA biogenesis. Open Biol. 2022 Jan;12(1):210305. doi: 10.1098/rsob.210305. Epub 2022 Jan 26. PMID: 35078352
|
 |
Marczyk M, Gunasekharan V, Casadevall D, Qing T, Foldi J, Sehgal R, Shan NL, Blenman KRM, O’Meara TA, Umlauf S, Surovtseva YV, Muthusamy V, Rinehart J, Perry RJ, Kibbey R, Hatzis C, Pusztai L. Comprehensive analysis of metabolic isozyme targets in cancer. Cancer Res. 2022 Feb 28;canres.CAN-21-3983-E.2021. doi: 10.1158/0008-5472.CAN-21-3983. PMID: 35247885
|
 |
Mohsen H, Gunasekharan V, Qing T, Seay M, Surovtseva Y, Negahban S, Szallasi Z, Pusztai L, Gerstein MB. Network propagation-based prioritization of long tail genes in 17 cancer types. Genome Biol. 2021 Oct 7;22(1):287. doi: 10.1186/s13059-021-02504-x. PMID: 34620211
|
 |
Wernke KM, Tirla A, Xue M, Surovtseva YV, Menges FS, Herzon SB. Probing Microbiome Genotoxicity: A Stable Colibactin Provides Insight into Structure-Activity Relationships and Facilitates Mechanism of Action Studies. J Am Chem Soc. 2021 Sep 29;143(38):15824-15833. doi: 10.1021/jacs.1c07559. PMID: 34524796 |
 |
King ALO, Mirza FN, Lewis JM, Umlauf S, Surovtseva Y, Carlson KR, Foss FM, Girardi M. Uncovering the potential of PI3K inhibitors in cutaneous T cell lymphoma: insights from high throughput in vitro screenings. J Invest Dermatol. 2021 Jul 19;S0022-202X(21)01451-2. doi: 10.1016/j.jid.2021.04.035. PMID: 34293349
|
 |
Sun Y, Abriola L, Niederer RO, Pedersen SF, Alfajaro MM, Monterio VS, Wilen CB, Ho Y, Gilbert WV, Surovtseva YV, Lindenbach BD, Guo JU. Restriction of SARS-CoV-2 replication by targeting programmed -1 ribosomal frameshifting. Proc Natl Acad Sci U S A. 2021 Jun 29; 118(26):e2023051118. doi: 10.1073/pnas.2023051118.
|
 |
Paradkar S, Herrington J, Hendricson A, Hewawasam P, Plummer M, Hoyer D, Sundaram RK, Surovtseva YV, Bindra RS. Creation of a new class of radiosensitizers for glioblastoma based on the mibefradil pharmacophore. Oncotarget. 2021 Apr 27;12(9):891-906. doi: 10.18632/oncotarget.27933.
|
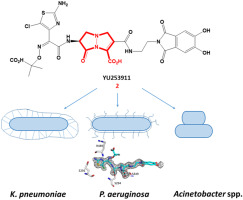 |
Goldberg JA, Kumar V, Spencer EJ, Hoyer D, Marshall SH, Hujer AM, Hujer KM, Bethel CR, Papp-Wallace KM, Perez F, Jacobs MR, van Duin D, Kreiswirth BN, van den Akker F, Plummer MS, Bonomo RA. A ɤ-lactam siderophore antibiotic effective against multidrug-resistant Pseudomonas aeruginosa, Klebsiella pneumoniae, and Acinetobacter spp. European Journal of Medicinal Chemistry. 2021 Apr 8. doi.org/10.1016/j.ejmech.2021.113436
|
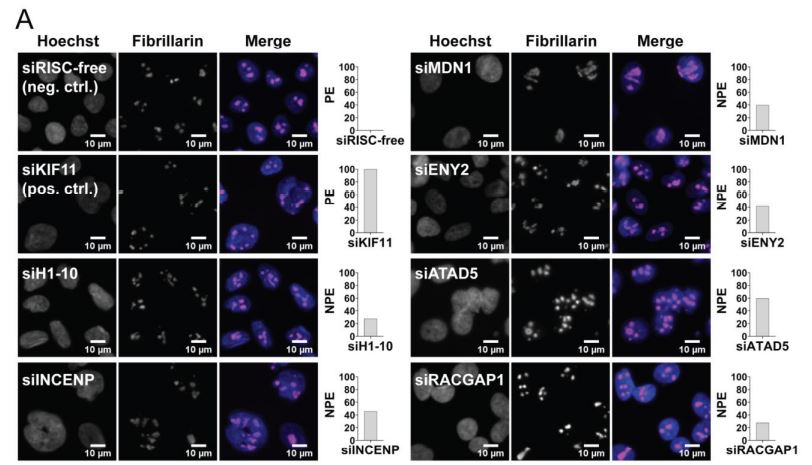 |
Ogawa LM, Buhagiar AF, Abriola L, Leland BA, Surovtseva YV, Baserga SJ. Increased numbers of nucleoi in a genome-wide RNAi screen reveal proteins that link the cell cycle to RNA polymerase I transcription. Mol Biol Cell. 2021 Mar 10; mbcE20100670. PMID: 33689394, DOI: 10.1091/mbc.E20-10-0670.
|
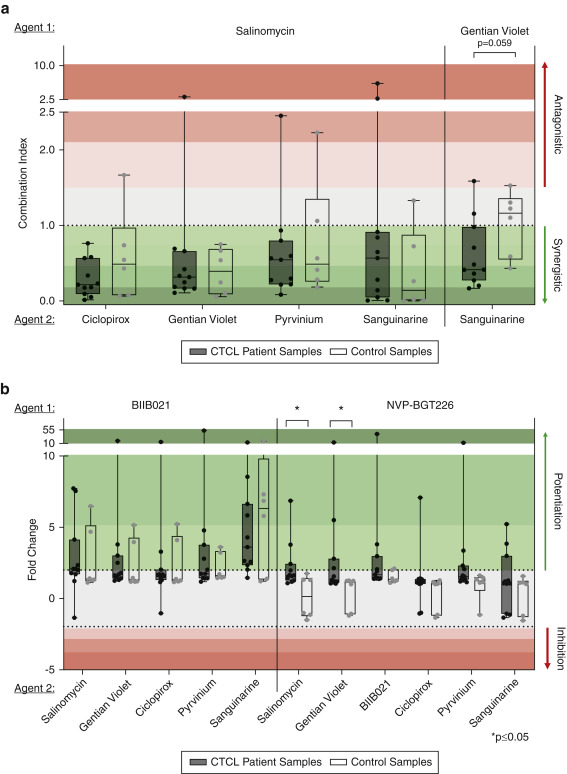 |
Mirza FN, Yumeen S, Lewis JM, King ALO, Kim SR, Carlson KR, Umlauf S, Surovtseva YV, Foss FM, Girardi M. Screening Novel Agent Combinations to Expedite CTCL Therapeutic Development. J Invest Dermatol. 2021 Jan;141(1):217-221. PMID: 32534802, DOI: 10.1016/j.jid.2020.05.097.
|
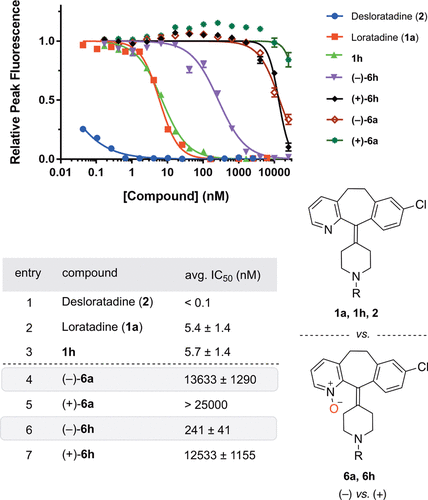 |
Stone EA, Cutrona KJ, Miller SJ. Asymmetric Catalysis upon Helically Chiral Loratadine Analogues Unveils Enantiomer-Dependent Antihistamine Activity. J Am Chem Soc. 2020 Jul 22;142(29):12690-12698. PMID: 32579347 DOI: 10.1021/jacs.0c03904 |
 |
Wei J, Alfajaro MM, Hanna RE, DeWeirdt PC, Strine MS, Lu-Culligan WJ, Zhang S, Graziano VR, Schmitz CO, Chen JS, Mankowski MC, Filler RB, Gasque V, de Miguel F, Chen H, Oguntuyo K, Abriola L, Surovtseva YV, Orchard RC, Lee B, Lindenbach B, Politi K, van Dijk D, Simon MD, Yan Q, Doench JG, Wilen CB. (2020) Genome-wide CRISPR screen reveals host genes that regulate SARS-CoV-2 infection. Cell 2020 Jun 17;2020.06.16.155101. doi: 10.1101/2020.06.16.155101. Preprint |
 |
Gannam ZTK , Min K, Shillingford SR, Zhang L, Herrington J, Abriola L, Gareiss PC, Pantouris G, Tzouvelekis A, Kaminski N, Zhang X, Yu J, Jamali H, Ellman JA, Lolis E, Anderson KS, Bennett AM. (2020) An allosteric site on MKP5 reveals a strategy for small-molecule inhibition. Sci Signaling 020 Aug 25;13(646):eaba3043. doi: 10.1126/scisignal.aba3043. PMID: 32843541
|
 |
Goldberg JA, Nguyen H, Kumar V, Spencer EJ, Hoyer D, Marshall EK, Cmolik A, O’Shea M, Marshall SH, Hujer AM, Hujer KM, Rudin SD, Domitrovic TN, Bethel CR, Papp-Wallace KM, Logan LK, Perez F, Jacobs MR, van Duin D, Kreiswirth BM, Bonomo RA, Plummer MS, van den Akker F. A γ-Lactam Siderophore Antibiotic Effective against Multidrug-Resistant Gram-Negative Bacilli. J Med Chem. 2020 Jun 11; 63(11):5990-6002. doi: 10.1021/acs.jmedchem.0c00255. PMID: 32420736
|
 |
Yumeen S., Mirza F.N., Lewis J.M., King A.L.O., Kim S.R., Carlson K.R., Umlauf S., Surovtseva Y.V., Foss F.M., Girardi M. (2020) JAK inhibition synergistically potentiates BCL2, BET, HDAC and proteasome inhibition in advanced CTCL. Accepted to Blood Advances. April 2020 PMID: 32437546, PMCID: PMC7252559, DOI: 10.1182/bloodadvances.2020001756.
|
 |
Rawling DC, Jagdmann GE, Potapova O, Pyle AM. Small-Molecule Antagonists of the RIG-I Innate Immune Receptor. ACS Chemical Biology. 2020 15 (2), 311-317. DOI: 10.1021/acschembio.9b00810.
|
 |
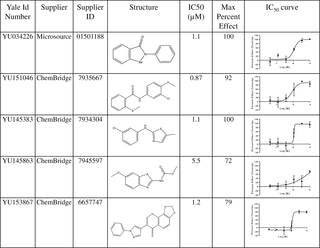
Perkins KR, Atilho RM, Moon MH, Breaker RR. Employing a ZTP Riboswitch to Detect Bacterial Folate Biosynthesis Inhibitors in a Small Molecule High-throughput Screen. ACS Chem Biol. 2019 Oct 14. [Epub ahead of print] doi: 10.1021/acschembio.9b00713. PMID:31609568. |
 |
Colavito SA, Platt JT, Held MA, Liu Z, Sokup R, Stern DF. Combinatorial drug screening of mammary cells with induced mesenchymal transformation to identify drug combinations for triple-negative breast cancer. Oncotarget. 2019 Aug 6;10(47):4822-4839. doi: 10.18632/oncotarget.27104. PMID: 31448050 PMCID: PMC6690678.
|
 |
Hendricson A, Umlauf S, Choi JY, Thekkiniath J, Surovtseva YV, Fuller KK, Reynolds TB, Voelker DR, Ben Mamoun C. High-throughput screening for phosphatidylserine decarboxylase inhibitors using a distyrylbenzene-bis-aldehyde (DSB-3)-based fluorescence assay. J Biol Chem. 2019 Aug 9;294(32):12146-12156. doi: 10.1074/jbc.RA119.007766. PMID: 31227523.
|
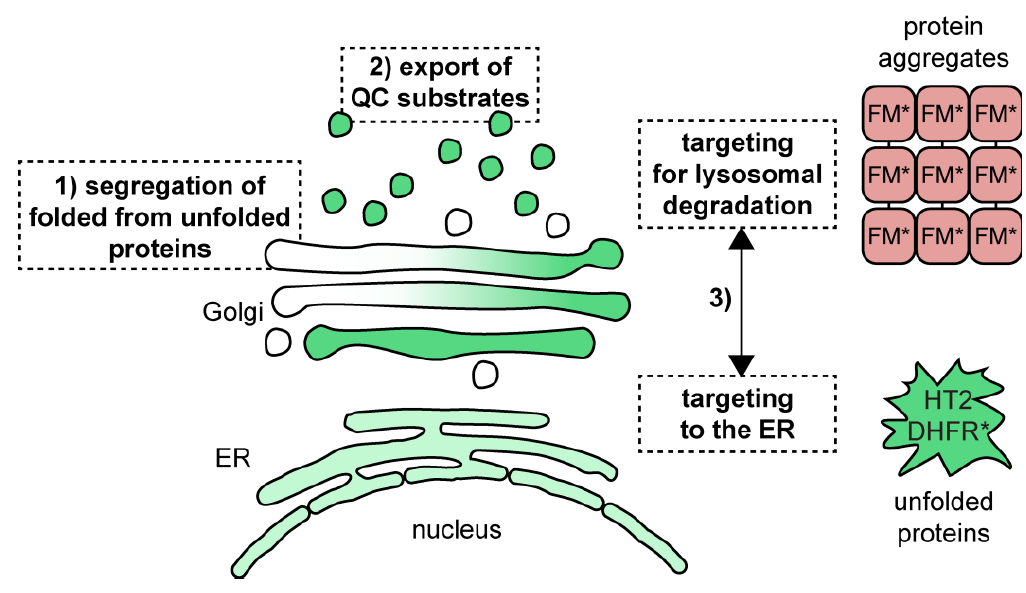 |
Hellerschmied D, Serebrenik YV, Shao L, Burslem GM, Crews CM. Protein Folding State-dependent Sorting at the Golgi Apparatus. Mol Biol Cell. 2019 Aug 1;30(17):2296-2308. doi.org/10.1091/mbc.E19-01-0069. PMID: 31166830.
|
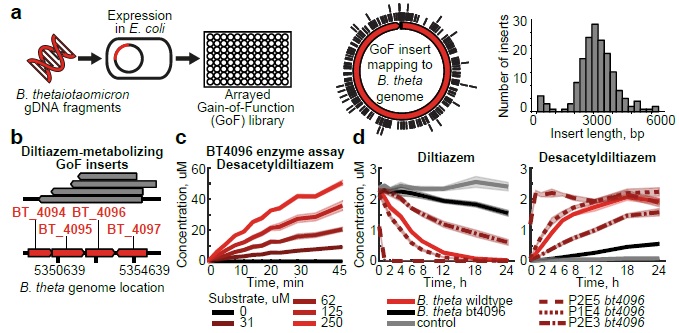 |
Zimmermann M, Zimmermann-Kogadeeva M, Wegmann R, Goodman AL. Mapping human microbiome drug metabolism by gut bacteria and their genes. Nature. 2019 Jun;570(7762):462-467. doi:10.1038/s41586-019-1291-3. PMID: 31158845. PMCID: PMC6597290.
|
 |
Abriola, L, Hoyer, D, Caffrey, CR, Williams, DL, Yoshino, TP, Vermeire, JJ. Development and optimization of a highthroughput screening method utilizing Ancylostoma ceylanicum egg hatching to identify novel anthelmintics. PLoS ONE 2019 June 3: 14(6): e0217019. doi.org/10.1371/journal.pone.0217019.
|
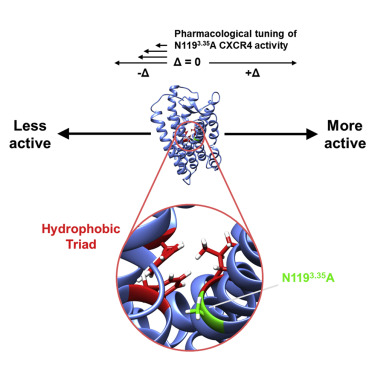 |
Rosenberg EM Jr, Harrison RES, Tsou LK, Drucker N, Humphries B, Rajasekaran D, Luker KE, Wu CH, Song JS, Wang CJ, Murphy JW, Cheng YC, Shia KS, Luker GD, Morikis D, Lolis EJ. Characterization, Dynamics, and Mechanism of CXCR4 Antagonists on a Constitutively Active Mutant. Cell Chem Biol. 2019 May 16: 26(5):662-673. doi: 10.1016/j.chembiol.2019.01.012. PMID: 30827936.
|
 |
Cai, H, Gong, J, Abriola, L, Hoyer, D, Stem Cell Array Team NG, Noggle, S, Paull, D, Del Priore, LV, Fields, MA. High-throughput screening identifies compounds that protect RPE cells from physiological stressors present in AMD. Exp Eye Res. 2019 Aug;185:107641. doi: 10.1016/j.exer.2019.04.009. PMID:30980814.
|
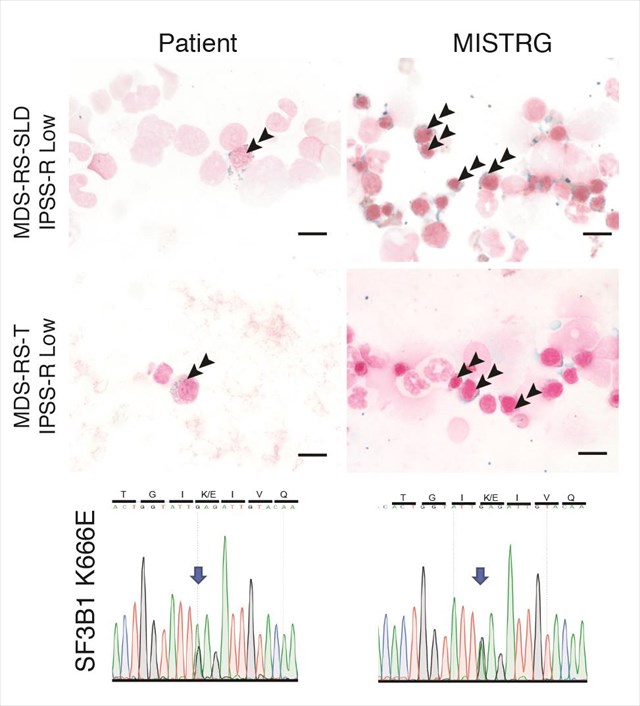 |
Song Y, Rongvaux A, Taylor A, Jiang T, Tebaldi T, Balasubramanian K, Bagale A, Terzi YK, Gbyli R, Wang X, Fu X, Gao Y, Zhao J, Podoltsev N, Xu M, Neparidze N, Wong E, Torres R, Bruscia EM, Kluger Y, Manz MG, Flavell RA, Halene S. A highly efficient and faithful MDS patient-derived xenotransplantation model for pre-clinical studies. Nature Communications. 2019 Jan 21; 10(1):366. PMID:30664659. PMCID: PMC6341122.
|
 |
Steinauer A, LaRochelle JR, Knox SL, Wissner RF, Berry S, Schepartz A. HOPS-dependent endosomal fusion required for efficient cytosolic delivery of therapeutic peptides and small proteins. Proc Natl Acad Sci U S A. 2019 Jan 8;116(2):512-521. doi: 10.1073/pnas.1812044116. PMID: 30610181. PMCID: PMC6329960.
|
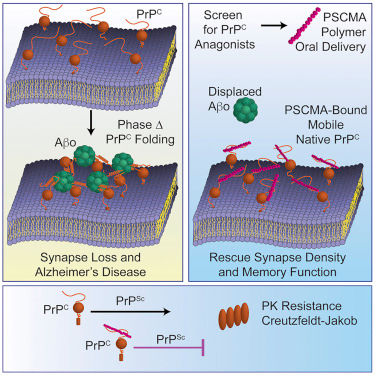 |
Gunther EC, Smith LM, Kostylev MA, Cox TO, Kaufman AC, Lee S, Folta-Stogniew E, Maynard GD, Um JW, Stagi M, Heiss JK, Stoner A, Noble GP, Takahashi H, Haas LT, Schneekloth JS, Merkel J, Teran C, Naderi ZK, Supattapone S, Strittmatter SM. Rescue of Transgenic Alzheimer’s Pathophysiology by Polymeric Cellular Prion Protein Antagonists. Cell Rep. 2019 Jan 2;26(1):145-158.e8. doi: 10.1016/j.celrep.2018.12.021. PMID: 30605671. PMCID: PMC658723. |
 |
Shine EE, Xue M, Patel JR, Healy AR, Surovtseva YV, Herzon SB, Crawford JM. Model Colibactins Exhibit Human Cell Genotoxicity in the Absence of Host Bacteria. ACS Chem Biol. 2018 Nov 20. doi: 10.1021/acschembio.8b00714. PMID: 30403848. PMCID: PMC5874909. |
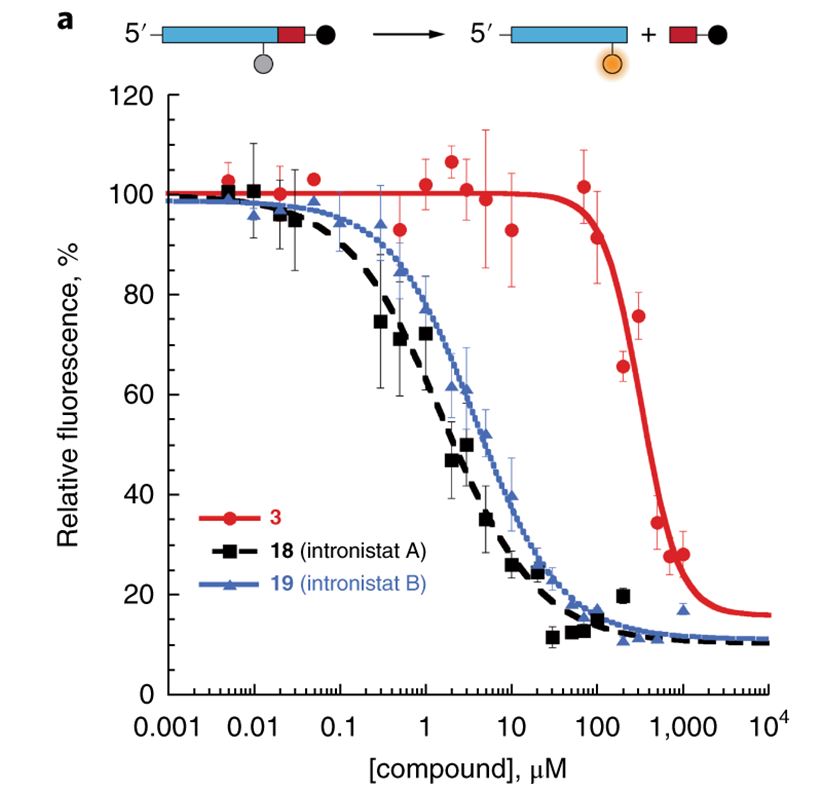 |
Fedorova O, Jagdmann Jr. GE, Adams RL, Yuan L, Van Zandt MC, Pyle AM. Small molecules that target group II introns are potent antifungal agents. Nat Chem Bio. 2018 Oct 15. PMID: 30323219. PMCID: PMC6239893.
|
 |
Huebner EA, Budel S, Jiang Z, Omura T, Ho TS, Barrett L, Merkel JS, Pereira LM, Andrews NA, Wang X, Singh B, Kapur K, Costigan M, Strittmatter SM, Woolf CJ. Diltiazem Promotes Regenerative Axon Growth. Mol Neurobiol. 2018 Sep 19. doi: 10.1007/s12035-018-1349-5. PMID: 30232777. PMCID: PMC6424671.
|
 |
Gassaway BM, Petersen MC, Surovtseva YV, Barber KW, Sheetz JB, Aerni HR, Merkel JS, Samuel VT, Shulman GI, Rinehart J. PKCε contributes to lipid-induced insulin resistance through cross talk with p70S6K and through previously unknown regulators of insulin signaling. Proc Natl Acad Sci U S A. 2018 Sep 18; 115(38):E8996-E9005. PMID: 30181290. PMCID: PMC6156646. |
 |
Wang G, Chow RD, Ye L, Guzman CD, Dai X, Dong MB, Zhang F, Sharp PA, Platt RJ, Chen S. Mapping a functional cancer genome atlas of tumor suppressors in mouse liver using AAV-CRISPR-mediated direct in vivo screening. Sci Adv. 2018 Feb 28;4(2). PMID: 29503867. PMCID: PMC5829971.
|
 |
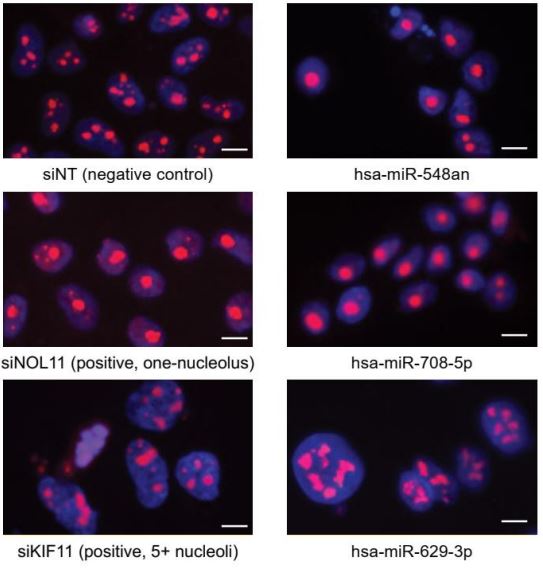
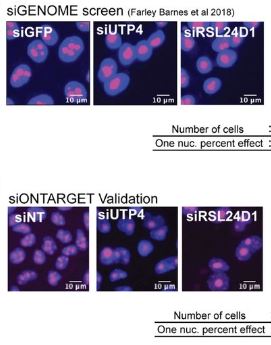
Farley-Barnes KI, McCann KL, Ogawa LM, Merkel J, Surovtseva YV, Baserga SJ. Diverse Regulators of Human Ribosome Biogenesis Discovered by Changes in Nucleolar Number. Cell Rep. 2018 Feb 13;22(7):1923-1934. doi: 10.1016/j.celrep.2018.01.056. PMID: 29444442. PMICD: PMC5828527.
Although ribosome biogenesis is an essential cellular process, many of the intricacies of its regulation in higher eukaryotes remain unknown. To understand how ribosome biogenesis is globally integrated in human cells, Baserga Lab collaborated with YCMD to conduct a genome-wide siRNA screen for regulators of nucleolar number. We found 139 proteins whose depletion changed the number of nucleoli per nucleus from 2–3 to only 1 in human MCF10A cells. Follow-up analyses on 20 hits found many (90%) to be essential for the nucleolar functions of rDNA transcription (7), pre-ribosomal RNA (pre-rRNA) processing (16), and/or global protein synthesis (14). This genome-wide analysis exploits the relationship between nucleolar number and function to discover diverse cellular pathways that regulate the making of ribosomes and paves the way for further exploration of the links between ribosome biogenesis and human disease.
|
 |
Ouyang X, Han SN, Zhang JY, Dioletis E, Nemeth BT, Pacher P, Feng D, Bataller R, Cabezas J, Stärkel P, Caballeria J, LePine Pongratz R, Cai SY, Schnabl B, Hoque R, Chen Y, Yang WH, Garcia-Martinez I, Wang FS, Gao B, Torok NJ, Kibbey RG, Mehal WZ. Digoxin Suppresses Pyruvate Kinase M2 Promoted HIF-1α Transactivation in Steatohepatitis. Cell Metabolism 2018 Feb 6;27(2):339-350.e3. doi: 10.1016/j.cmet.2018.01.007. PMID: 29414684. PMCID: PMC5806149.
|
 |
Choi JY, Surovtseva YV, Van Sickle SM, Kumpf J, Bunz UHF, Ben Mamoun C, Voelker DR. A novel fluorescence assay for measuring phosphatidylserine decarboxylase catalysis. J Biol Chem. 2018 Feb 2;293(5):1493-1503. doi: 10.1074/jbc.RA117.000525. PMID: 29247006. PMCID: PMC5798280.
|
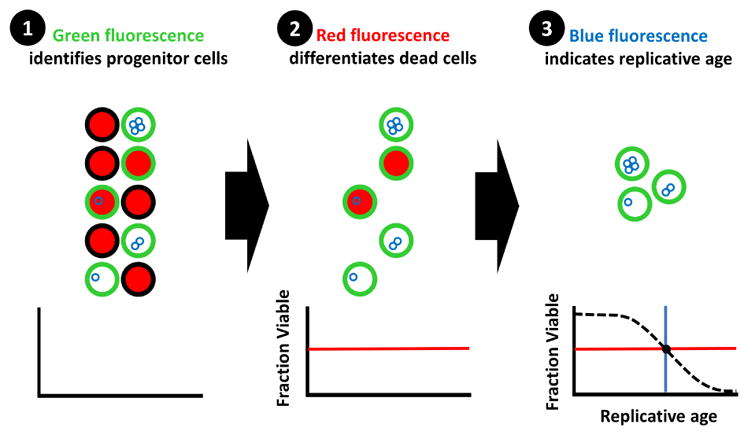 |
Sarnoski EA, Liu P, Acar M. A High-Throughput Screen for Yeast Replicative Lifespan Identifies Lifespan-Extending Compounds. Cell Rep. 2017 Nov 28;21(9):2639-2646. doi: 10.1016/j.celrep.2017.11.002. PMID: 29186697. PMCID: PMC5728442.
|
 |
Miller CJ, Muftuoglu Y, Turk BE. A high throughput assay to identify substrate-selective inhibitors of the ERK protein kinases. Biochem Pharmacol. 2017 Oct 15;142:39-45. doi: 10.1016/j.bcp.2017.06.127. PMID: 28647489. PMCID: PMC5678991.
|
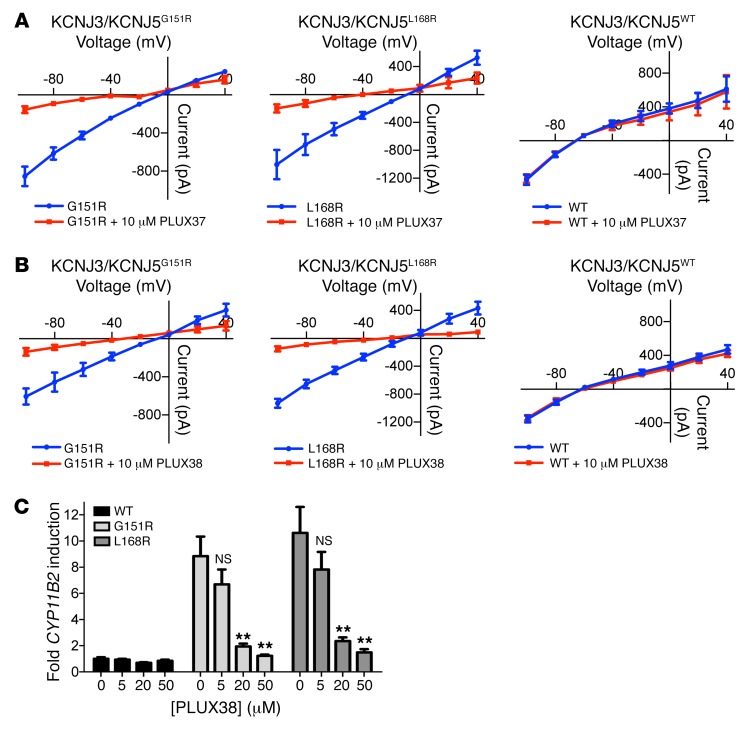 |
Scholl UI, Abriola L, Zhang C, Reimer EN, Plummer M, Kazmierczak BI, Zhang J, Hoyer D, Merkel JS, Wang W, Lifton RP: Macrolides selectively inhibit mutant KCNJ5 potassium channels that cause aldosterone-producing adenoma. J Clin Invest. 2017 Jun 30; 127(7):2739-2750. doi: 10.1172/JCI91733. PMID: 28604387. PMCID: PMC5490757.
Mutations in the potassium channel KCNJ5 (hereafter referred to as KCNJ5MUT) are associated with Aldosterone-producing adenomas (APAs) - benign tumors of the adrenal gland that cause millions of cases of severe hypertension worldwide. Because APA diagnosis requires a difficult invasive procedure, patients often remain undiagnosed and inadequately treated. Inhibitors of KCNJ5MUT could allow noninvasive diagnosis and therapy of APAs carrying KCNJ5 mutations. Here, Lifton lab collaborated with YCMD to develop a high-throughput screen for rescue of KCNJ5MUT-induced lethality, and identify a series of macrolide antibiotics, including roxithromycin, that potently inhibit KCNJ5MUT, but not KCNJ5WT. Macrolide-derived selective KCNJ5MUT inhibitors thus have the potential to advance the diagnosis and treatment of APAs harboring KCNJ5MUT.
|
 |
Langdon CG, Platt JT, Means RE, Iyidogan P, Mamillapalli R, Klein M, Held MA, Lee JW, Koo JS, Hatzis C, Hochster HS, Stern DF. Combinatorial screening of pancreatic adenocarcinoma reveals sensitivity to drug combinations including bromodomain inhibitor plus neddylation inhibitor. Mol Cancer Ther. 2017 Jun;16(6):1041-1053. doi: 10.1158/1535-7163.MCT-16-0794. PMID: 28292938. PMCID: PMC5457712.
Pancreatic adenocarcinoma (PDAC) is the fourth most common cause of cancer-related death in the United States. PDAC is largely driven by activating KRAS mutations, and as such, cannot be directly targeted with therapeutic agents that affect the activated protein. Instead, inhibition of downstream signaling and other targets will be necessary to effectively manage PDAC. Here, Stern laboratory collaborated with YCMD to perform a tiered single-agent and combination compound screen to identify targeted agents that impair growth of a panel of PDAC cell lines. Several of the combinations identified from the screen were further validated for efficacy and mechanism. Overall, this work revealed novel combinatorial regimens, including JQ1 plus MLN4294, which show promise for the treatment of RAS-driven PDAC.
|
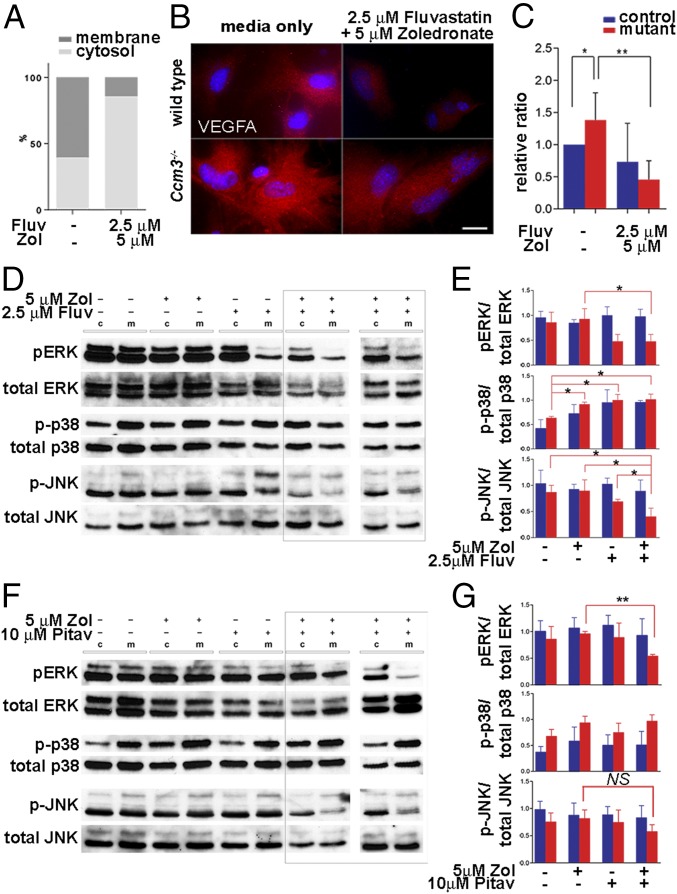 |
Nishimura S, Mishra-Gorur K, Park J, Surovtseva YV, Sebti SM, Levchenko A, Louvi A, Gunel M. Combined HMG-COA reductase and prenylation inhibition in treatment of CCM. Proc Natl Acad Sci U S A. 2017 May 23;114(21):5503-5508. doi: 10.1073/pnas.1702942114. PMID: 28500274. PMCID: PMC5448170.
Cerebral cavernous malformations (CCMs) are common vascular anomalies that develop in the central nervous system and, more rarely, the retina. Here, Gunel lab collaborated with YCMD to optimize and implement a high-throughput screen to identify bioactive compounds that could effectively suppress hyperproliferation of mouse brain primary astrocytes deficient for CCM3. Gunel lab demonstrated that screen actives fluvastatin, an inhibitor of 3-hydroxy-3-methyl-glutaryl (HMG)-CoA reductase and the N-bisphosphonate zoledronic acid monohydrate, an inhibitor of protein prenylation, act synergistically to reverse outcomes of CCM3 loss in cultured mouse primary astrocytes and in Drosophila glial cells in vivo. Further, the two drugs effectively attenuate neural and vascular deficits in chronic and acute mouse models of CCM3 loss in vivo, providing a potential pharmacological treatment option for CCM disease.
|
 |
Puleo DE, Kucera K, Hammeren HM, Ungureanu D, Newton AS, Silvennoinen O, Jorgensen WL, Schlessinger J. Identification and Scharacterization of JAK2 Pseudokinase Domain Small Molecule Binders. ACS Med Chem Lett. 2017 May 17;8(6):618-621. doi: 10.1021/ascmedchemlett. 7b00153. PMID:28626521. PMCID:PMC5467198. |
 |
Thakral D, Tae HS. Discovery of a Structurally Unique Small Molecule that Inhibits Protein Synthesis. Yale J Biol Med. 2017 Mar 29;90(1):35-43. PMID: 28356892. PMCID: PMC5369043.
|
 |
Nemec AA, Abriola L, Merkel JS, deStanchina E, DeVeaux M, Zelterman D, Glazer PM, Sweasy JB. DNA polymerase beta germline variant confers cellular response to cisplatin therapy. Mol Cancer Res. 2017 Mar;15(3):269-280. doi: 10.1158/1541-7786.MCR-16-0227-T. PMID: 28074003. PMCID: PMC5334281.
Resistance to cancer chemotherapies leads to deadly consequences. This paper focused on a P242R mutation in DNA polymerase beta (Pol β) which is associated with poor prognosis for lung cancer, specifically in response to treatment with cisplatin. Here, Glazer and Sweasy laboratories collaborated with YCMD to show that the P242R mutation is sufficient to promote resistance to cisplatin in human cells. This phenotype was also validated in vivo in mouse xenografts. Importantly, in a mouse xenograft model, tumors derived from lung cancer cells expressing WT Pol β displayed a slower rate of growth when treated with cisplatin, whereas tumors expressing P242R Pol β had no response to cisplatin. The current data strongly suggests that the status of Pol β influences cellular responses to crosslinking agents and that Pol β is a promising biomarker to predict responses to specific chemotherapies.
|
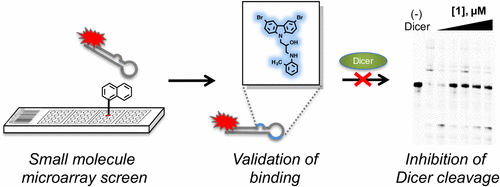 |
Connelly CM, Boer RE, Moon MH, Gareiss P, Schneekloth JS Jr. Discovery of Inhibitors of MicroRNA-21 Processing Using Small Molecule Microarrays. ACS Chem Biol. 2017 Feb 17;12(2):435-443. doi: 10.1021/acschembio.6b00945. PMID: 27959491. PMCID: PMC6341489.
|
 |
Sulkowski PL, Corso CD, Robinson ND, Scanlon SE, Purshouse KR, Bai H, Liu Y, Sundaram RK, Hegan DC, Fons NR, Breuer GA, Song Y, Mishra-Gorur K, De Feyter HM, de Graaf RA, Surovtseva YV, Kachman M, Halene S, Günel M, Glazer PM, Bindra RS. 2-Hydroxyglutarate produced by neomorphic IDH mutations suppresses homologous recombination and induces PARP inhibitor sensitivity. Sci Transl Med. 2017 Feb 1;9(375). pii: eaal2463. doi: 10.1126/scitranslmed.aal2463. PMID: 28148839. PMCID: PMC5435119.
2-Hydroxyglutarate (2HG) is implicated in tumor progression via its inhibitory effects on α-ketoglutarate (αKG)-dependent dioxygenases. This paper reports that IDH1/2 mutations induce a homologous recombination (HR) defect that renders tumor cells exquisitely sensitive to poly(adenosine 5’-diphosphate-ribose) polymerase (PARP) inhibitors. This “BRCAness” phenotype of IDH mutant cells can be completely reversed by treatment with small-molecule inhibitors of the mutant IDH1 enzyme, and conversely, it can be entirely recapitulated by treatment with either of the 2HG enantiomers in cells with intact IDH1/2 proteins. Bindra, Glazer and Gunel labs demonstrated mutant IDH1-dependent PARP inhibitor sensitivity in a range of clinically relevant models, including primary patient-derived glioma cells in culture and genetically matched tumor xenografts in vivo. YCMD contributed by developing and implementing image-based protocols to detect and quantify DNA damage foci in cell cycle dependent manner.
|
 |
Kraehling JR, Chidlow JH, Rajagopal C, Sugiyama MG, Fowler JW, Lee MY, Zhang X, Ramírez CM, Park EJ, Tao B, Chen K, Kuruvilla L, Larriveé B, Folta-Stogniew E, Ola R, Rotllan N, Zhou W, Nagle MW, Herz J, Williams KJ, Eichmann A, Lee WL, Fernández-Hernando C, Sessa WC. Genome-wide RNAi screen reveals ALK1 mediates LDL uptake and transcytosis in endothelial cells. Nat Commun. 2016 Nov 21;7:13516. doi: 10.1038/ncomms13516. PMID: 27869117. PMCID: PMC5121336.
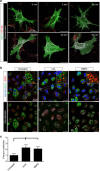
In humans and animals lacking functional LDL receptor (LDLR), LDL from plasma still readily traverses the endothelium. To identify the pathways of LDL uptake, a genome-wide RNAi screen was performed in endothelial cells and cross-referenced with GWAS-data sets. Here we show that the activin-like kinase 1 (ALK1) mediates LDL uptake into endothelial cells. ALK1 binds LDL with lower affinity than LDLR and saturates only at hypercholesterolemic concentrations. ALK1 mediates uptake of LDL into endothelial cells via an unusual endocytic pathway that diverts the ligand from lysosomal degradation and promotes LDL transcytosis. The endothelium-specific genetic ablation of Alk1 in Ldlr-KO animals leads to less LDL uptake into the aortic endothelium, showing its physiological role in endothelial lipoprotein metabolism. In summary, identification of pathways mediating LDLR-independent uptake of LDL may provide unique opportunities to block the initiation of LDL accumulation in the vessel wall or augment hepatic LDLR-dependent clearance of LDL. |
 |
Weicksel SE, Mahadav A, Moyle M, Cipriani PG, Kudron M, Pincus Z, Bahmanyar S, Abriola L, Merkel J, Gutwein M, Fernandez AG, Piano F, Gunsalus KC, Reinke V. A novel small molecule that disrupts a key event during the oocyte-to-embryo transition in C. elegans. Development. 2016 Oct 1;143(19):3540-3548. PMID: 27510972. PMCID: PMC5087616. |
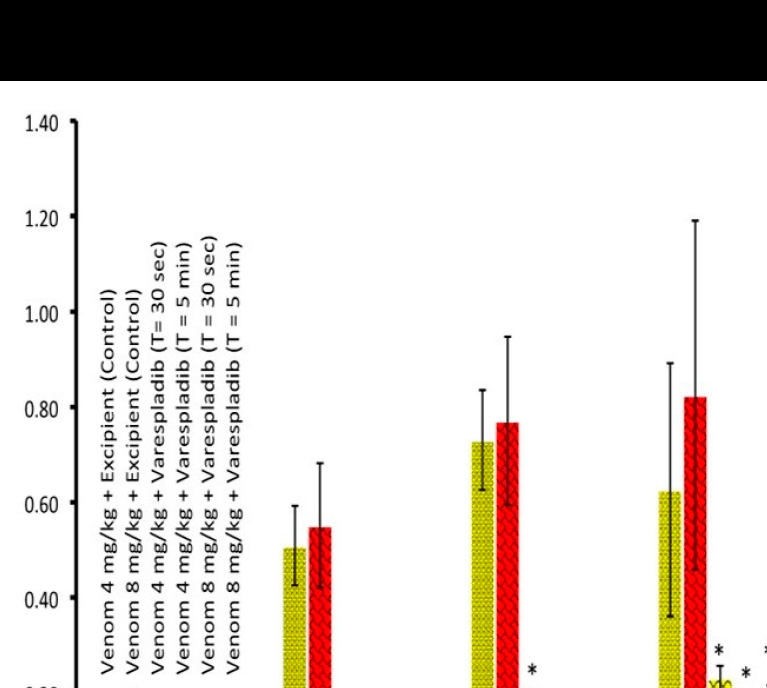 |
Lewin, M, Samuel, S, Merkel, J, Bickler, P. Varespladib (LY315920) appears to be a potent, broad-spectrum, inhibitor of snake venom phospholipase A2 and a possible pre-referral treatment for envenomation. Toxins 2016 Aug 25, 8(9), 248; doi:10.3390/toxins8090248. PMID:27571102. PMCID: PMC5037474.
Working with external groups, YCMD optimized an enzymatic assay for soluble phospholipase A2 activity across 28 medically important snake venoms from six continents. Two related inhibitors were demonstrated to powerfully inhibit venoms displaying nanomolar and subnanomolar activity. Inhibitor testing preceded animal studies showing a survival benefit when the small molecule inhibitors were added before or after venom exposure.
|
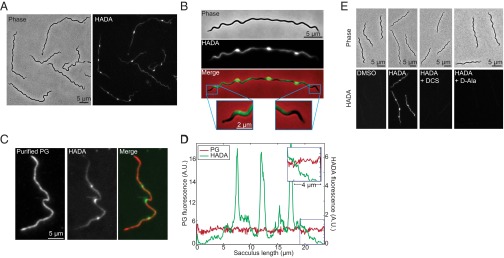 |
Jutras BL, Scott M, Parry B, Biboy J, Gray J, Vollmer W, Jacobs-Wagner C. Lyme disease and relapsing fever Borrelia elongate through zones of peptidoglycan synthesis that mark division sites of daughter cells. Proc Natl Acad Sci U S A. 2016 Aug 16;113(33):9162-70. doi: 10.1073/pnas.1610805113. PMID: 27506799. PMCID: PMC4995948. |
 |
Gale M, Sayegh J, Cao J, Norcia M, Gareiss P, Hoyer D, Merkel JS, Yan Q. Screen-identified selective inhibitor of lysine demethylase 5A blocks cancer cell growth and drug resistance. Oncotarget. Vol. 5. 2016 Jun 28;7(26):39931-39944; doi: 10.18632/oncotarget.9539. PMID: 27224921. PMCID: PMC5129982.
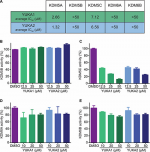
Lysine demethylase 5A (KDM5A/RBP2/JARID1A) is a histone lysine demethylase that is overexpressed in several human cancers including lung, gastric, breast and liver cancers. It plays key roles in important cancer processes including tumorigenesis, metastasis, and drug tolerance, making it a potential cancer therapeutic target. Chemical tools to analyze KDM5A demethylase activity are extremely limited as available inhibitors are not specific for KDM5A.
The Yan lab and YCMD characterized KDM5A using a homogeneous luminescence-based assay and conducted a high throughput screen of ~ 9,000 small molecules for inhibitors. Several compounds were identified that inhibited KDM5A with low μM in vitro IC50 values. Importantly, these compounds showed specificity and did not inhibit its close homologue KDM5B or the related H3K27 demethylases KDM6A and KDM6B. One compound, was able to increase H3K4me3 levels in human cells and selectively inhibit the proliferation of cancer cells whose growth depends on KDM5A. This compound also hindered the emergence of drug-tolerence in EGFR-mutant lung cancer cells treated with gefitinib and HER2+ breast cancer cells treated with trastuzumab, highlighting the critical role of KDM5A demethylase activity in drug resistance. These small molecules identified from screening efforts in collaboration with YCMD are excellent tool compounds for further study of KDM5A’s demethylase activity and its contributions to cancer.
|
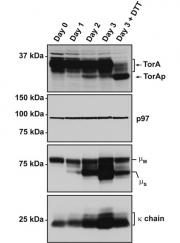 |
Zhao, C, Brown, R, Tang, C, Hu, C, Schlieker C. (2016), Site-specific Proteolysis Mobilizes TorsinA from the Membrane of the Endoplasmic Reticulum in Response to ER Stress and B cell Stimulation. J. Biol. Chem. 2016 Apr 29;291(18):9469-81 jbc.M115.709337. doi:10.1074/jbc.M115.709337. PMID: 26953341. PMCID: PMC:4850287.
Torsin ATPases are the only representatives of the AAA+ ATPase family that reside in the lumen of the endoplasmic reticulum (ER) and nuclear envelope. Two of these, TorsinA and TorsinB, are anchored to the ER membrane by virtue of an N-terminal hydrophobic domain. Here we demonstrate that the imposition of ER stress leads to a proteolytic cleavage event that selectively removes the hydrophobic domain from the AAA+ domain of TorsinA, which retains catalytic activity. Both the pharmacological inhibition profile and the identified cleavage site between two juxtaposed cysteine residues are distinct from those of presently known proteases, suggesting that a hitherto uncharacterized, membrane-associated protease accounts for TorsinA processing. This processing occurs not only in stress-exposed cell lines but also in primary cells from distinct organisms including stimulated B cells, indicating that Torsin conversion in response to physiologically relevant stimuli is an evolutionarily conserved process. By establishing 5-nitroisatin as cell-permeable inhibitor for Torsin processing, we provide the methodological framework for interfering with Torsin processing in a wide range of primary cells without the need for genetic manipulation.
|
 |
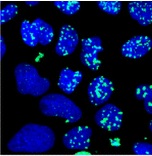
Li S, Hwang XY, Stav S, Breaker RR. The yjdF riboswitch candidate regulates gene expression by binding diverse azaaromatic compounds. RNA. 2016 Apr;22(4):530-41. doi: 10.1261/rna.054890.115. PMID: 26843526. PMCID: PMC4793209. |
 |
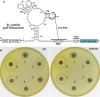
Surovtseva, Y, Jairam, V, Salem, AF, Bindra, RS, Herzon SB., Characterization of Cardiac Glycoside Natural Products as Potent Inhibitors of DNA Double-Strand Break Repair by a Whole-Cell Double Immunofluorescence Assay. J AM Chem Soc. 2016 Mar 23;138(11):3844-55. doi: 10.1021/jacs.6b00162. PMID: 26927829. PMCID: PMC5530877.
DNA repair pathways are being intensively investigated as targets for chemotherapeutic interventions. This paper describes development of an automated, whole-cell, high-throughput assay for the unbiased discovery of inhibitors of the non-homologous end joining (NHEJ) and homologous recombination (HR) repair pathways, the two primary pathways that ameliorate DNA double-strand breaks. The assay enables indirect measurement of DNA repair activity by monitoring the kinetics of γH2AX and 53BP1 foci resolution after treatment with ionizing radiation (IR) and candidate DNA damage response (DDR) inhibitor. The paper also describes a unique platform of secondary assays to rank-order the potency of lead compounds and gain insights into their mechanism of action. The HTS assay was validated using known DDR and DNA damage checkpoint inhibitors, and applied to screen 2,366 structurally-diverse small molecules with known bioactivity. YCMD contributed to the publication by optimizing a high-throughput assay, conducting a screen, and running follow up assays which identified cardiac glycoside natural products as potent Inhibitors of DNA Double-Strand Break Repair.
|
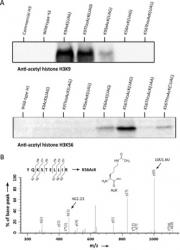 |
Xiong, H, Reynolds, NM, Fan, C, Englert, M, Hoyer, D, Miller, SJ and Söll, D. Dual Genetic Encoding of Acetyl-lysine and Non-deacetylatable Thioacetyl-lysine Mediated by Flexizyme. Angew. Chem. Int. Ed. 2016 Mar 14;55(12):4083-6. doi:10.1002/anie.201511750. PMID: 26914285. PMCID: PMC4789153.
Acetylation of lysine residues is an important post-translational protein modification. Lysine acetylation in histones and its crosstalk with other post-translational modifications in histone and non-histone proteins are crucial to DNA replication, DNA repair, and transcriptional regulation. We incorporated acetyl-lysine (AcK) and the non-hydrolyzable thioacetyl-lysine (ThioAcK) into full-length proteins in vitro, mediated by flexizyme. ThioAcK and AcK were site-specifically incorporated at different lysine positions into human histone H3, either individually or in pairs. We demonstrate that the thioacetyl group in histone H3 could not be removed by the histone deacetylase sirtuin type 1. This method provides a powerful tool to study protein acetylation and its role in crosstalk between post-translational modifications.
|
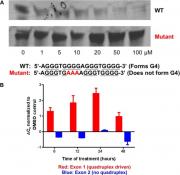 |
Felsenstein KM, Saunders LB, Simmons JK, Leon E, Calabrese DR, Zhang S, Michalowski A, Gareiss P, Mock BA, Schneekloth JS Jr. Small Molecule Microarrays Enable the Identification of a Selective, Quadruplex-Binding Inhibitor of MYC Expression. ACS Chem Biol. 2016 Jan 15;11(1):139-48. PMCID: PMC4719142. PMID: 26462961.
The transcription factor MYC plays a pivotal role in cancer initiation, progression, and maintenance. However, it has proven difficult to develop small molecule inhibitors of MYC. YCMD employed small molecule microarray technology to screen for compounds that bind to oncogenic MYC G-Quadruplex and potentially target gene expression. Compounds were identified that bind to this structured DNA target and affect gene expression. As a measure of selectivity, gene expression analysis and qPCR experiments demonstrated that MYC and several MYC target genes were downregulated upon treatment with this compound, while the expression of several other G-quadruplex driven genes was not affected. Follow-up studies with scientists at the NCI (former YCMD scientist Jay Schneekloth and colleagues) showed that this compound is able to inhibit MYC induced cellular toxicity.
|
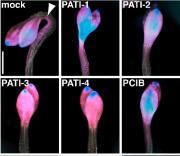 |
Carland F, Defries A, Cutler S, Nelson T. Novel Vein Patterns in Arabidopsis Induced by Small Molecules. Plant Physiol. 2016 Jan; 170(1):338-53. doi: 10.0004/pp.15.01540. PMID: 26574596. PMCID: PMC4704596.
Leaf veins play a critical role in transporting water, nutrients, and signals. To identify regulators of vein patterning, Carland et al. screened more than 5000 structurally diverse small molecules for compounds that alter Arabidopsis thaliana leaf vein patterns. High throughput methods developed at YCMD included robotic dispensing of agarose into 96 well-plates and optimization of seed loading for a consistent one seed per well.
|
 |
Langdon CG, Wiedemann N, Held MA, Mamillapalli R, Iyidogan P, Theodosakis N, Platt JT, Levy F, Vuagniaux G, Wang S, Bosenberg MW, Stern DF. SMAC mimetic Debio 1143 synergizes with taxanes, topoisomerase inhibitors and bromodomain inhibitors to impede growth of lung adenocarcinoma cells. Oncotarget. 2015 Nov 10;6(35):37410-25. PMID: 26485762. PMCID: PMC4741938.
|
 |
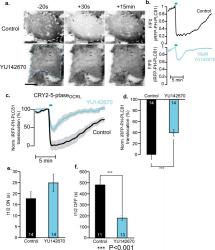
Stachelek GC, Peterson-Roth E, Liu Y, Fernandez RJ 3rd, Pike LR, Qian JM, Abriola L, Hoyer D, Hungerford W, Merkel J, Glazer PM. YU238259 is a Novel Inhibitor of Homology-dependent DNA Repair that Exhibits Synthetic Lethality and Radiosensitization in Repair-deficient Tumors. Mol Cancer Res. 2015 Oct; 13(10):1389-97. doi:10.1157/1541-7786.MCR-15-0036 PMID:26116172. PMCID: PMC4618103.
DNA repair pathways are potential targets for cancer therapy strategies. This paper describes a high throughput cell-based screen for small molecules that were preferentially toxic to cells lacking the FANCD2 and BRCA2 repair proteins, but had low toxicity in isogenic, repair-proficient cells. Work performed at YCMD included development of the synthetic lethal assay, high throughput screening, hit follow-up by the synthetic lethal assay, chemistry-based analysis of the hits and assessment of structure activity relationships (SAR), hit expansion by catalog and new molecule design and synthesis to improve the potency and solubility of the lead molecule. A novel series of compounds that are synthetically lethal to HDR-deficient cells was identified.
|
 |
Blount KF, Megyola C, Plummer M, Osterman D, O’Connell T, Aristoff P, Quinn C, Chrusciel RA, Poel TJ, Schostarez HJ, Stewart CA, Walker DP, Wuts PGM, Breaker RR. Novel riboswitch-binding flavin analog that protects mice against Clostridium difficile infection without inhibiting cecal flora. Antimicrob Agents Chemother 2015 Sept; 59:5736–5746. doi:10.1128/AAC.01282-15. PMID: 26169403. PMCID: PMC4538501. |
 |
Jane-Wit D, Surovtseva YV, Qin L, Li G, Liu R, Clark P, Manes TD, Kashgarian M, Kirkiles-Smith NC, Tellides G, Pober J. Complement membrane attack complexes activate noncanonical NF–kB by forming an Akt+NIK+ signalosome on Rab5+ endosomes. Proc Natl Acad Sci USA, 2015 Aug 4;112(31):9686-91. doi:10.1073/pnas. 1503535112. PMID: 21695760. PMCID: PMC4534258.
The paper investigates the mechanism of non-canonical NF-kB pathway activation by complement membrane attack complexes (MAC). The study elucidates important steps of pathway activation, describes the role of clathrin-mediated endocytosis and Rab5+ endosomes in MAC signaling, and has important translational implications by suggesting new therapeutic targets for controlling complement-mediated inflammation. YCMD contributed to the publication by optimizing a high-throughput assay and conducting a genome-wide siRNA screen, which identified endocytosis and vesicular trafficking genes as important players in the MAC-induced non-canonical NF-kB pathway activation.
|
 |
Langdon, CG, Held, MA, Platt, JT, Meeth, K, Iyidogan, P, Mamillapalli, R, Koo, AB, Klein, M, Liu, Z, Bosenberg, MW and Stern, DF. The broad-spectrum receptor tyrosine kinase inhibitor dovitinib suppresses growth of BRAF-mutant melanoma cells in combination with other signaling pathway inhibitors. Pigment Cell & Melanoma Research. 2015 Jul; 28(4):417-30. doi: 10.1111/pcmr.12376. PMID: 25854919. PMCID: PMC5215495.
While BRAF inhibitors have revolutionized treatment of mutant BRAF metastatic melanomas, resistance develops rapidly. Treatment with dovitinib in combination with several agents inhibits cell growth more effectively than either agent alone. Inhibition of BRAF-mutant melanoma and colorectal carcinoma cell growth was measured after incubation with combinations of dovitinib with other small molecules in a screen at YCMD. This work highlights the potential effectiveness of dovitinib with second agents to improve BRAF-mutant melanoma treatment, regardless of their sensitivity to BRAF inhibitors.
|
 |
Nath A, Schlamadinger DE, Rhoades E, Miranker AD. Structure-Based Small Molecule Modulation of a Pre-Amyloid State: Pharmacological Enhancement of IAPP Membrane-Binding and Toxicity. Biochemistry. 2015 Jun 9;54(22):3555-64. doi: 10.1021/acs.biochem.5b00052. PMID: 25966003. PMCID: PMC4697932. |
 |
Nelson JW, Plummer MS, Blount KF, Ames TD, Breaker RR. Small molecule fluoride toxicity agonists. Chem Biol. 2015 Apr 23;22(4):527-34. doi: 10.1016/j.chembiol.2015.03.016. PMID: 25910244. PMCID: PMC4617673.
Fluoride is a ubiquitous anion that inhibits a wide variety of metabolic processes. Here, we report the identification of a series of compounds that enhance fluoride toxicity in Escherichia coli and Streptococcus mutans. These molecules were isolated by using a high-throughput screen (HTS) at YCMD for compounds that increase intracellular fluoride levels as determined via a fluoride riboswitch reporter fusion construct. A series of derivatives were synthesized at YCMD to examine structure-activity relationships, leading to the identification of compounds with improved activity.
|
 |
Narayan A, Bommakanti A, Patel AA. High-throughput RNA profiling via up-front sample parallelization. Nat Methods. 2015 Apr; 12(4):343-6. doi: 10.1038/nmeth.3311b. PMCID: PMC4451056.
Analysis of gene expression within diverse specimens underpins our understanding of cellular physiology and disease. The Patel laboratory utilized the liquid handling infrastructure at YCMD to array microRNA to allow for an efficient platform for META RNA profiling.
|
 |
Scortegagna M, Lau E, Zhang T, Feng Y, Sereduk C, Yin H, De SK, Meeth K, Platt JT, Langdon CG, Halaban R, Pellecchia M, Davies MA, Brown K, Stern DF, Bosenberg M, Ronai ZA. PDK1 and SGK3 Contribute to the Growth of BRAF-Mutant Melanomas and Are Potential Therapeutic Targets. Cancer Res. 2015 Apr 1;75(7):1399-412. doi: 10.1158/0008-5472.CAN-14-2785. PMID: 25712345. PMCID: PMC4383687.
Recent Braf::Pten mutant studies have shown PDK1 to be involved in melanoma development. Interactions between PDKi (a PDK1 inhibitor) and 45 other agents were determined in a “one versus many” screen at YCMD. Specifically, melanoma cell growth was measured after incubation with the PKDi combinations. This work highlights the potential significance of PDK1 as a therapeutic target to improve melanoma treatment.
|
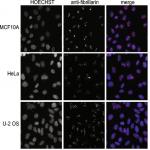 |
Farley KI, Surovtseva Y, Merkel J, Baserga SJ. Determinants of mammalian nucleolar architecture. Chromosoma. 2015 Feb 12. doi: 10.1007/s00412-015-0507-z. PMID: 25670395. PMCID: PMC4534358.
The nucleolus is a highly dynamic organelle with a complex structure. In this paper, nucleolar architecture was assayed at YCMD using a panel of mammalian cell lines. Specifically, nucleoli were immunostained using anti-fibrillarin antibodies and imaged on InCell2200, and their number and morphology was quantified using Cell Profiler pipeline developed at YCMD. We showed that nucleolar number varies greatly among mammalian cells, demonstrating the complexity of processes governing nucleolar development.
|
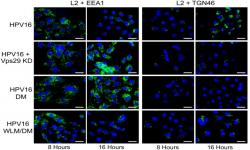 |
Popa A, Zhang W, Harrison M, Goodner K, Goodwin EC, Kazakov T, Burd C, DiMaio, D. Direct binding of the retromer to human papillomavirus 16 minor capsid protein L2 mediates endosome exit during virus entry. PLoS Pathogens. 2015 Feb 18;11(2). doi: 10.1371/journal.ppat.1004699. PMID: 25693203. PMCID: PMC4334968.
This paper presents follow up studies performed on hits identified in an siRNA screen conducted at YCMD.
|
 |
Schlamadinger D, Miranker A. Fiber-Dependent and Independent Toxicity of Islet Amyloid Polypeptide. Biophysical Journal. 2014 Dec;107(11):2559-2566. PMCID: PMC4255681.
The 37-residue peptide hormone islet amyloid polypeptide (IAPP) plays a central role in diabetes pathology. This paper describes cell-based toxicity experiments which directly assess amyloid fiber aggregation kinetics and cytotoxicity to β-cells, and shows that amyloid formation of IAPP can be strongly inhibited by the extracellular environment of live cells. YCMD staff has helped to develop some of the cell-based assays described in the paper.
|
 |
Hebda JA, Magzoub M, Miranker AD. Small molecule screening in context: Lipid-catalyzed amyloid formation. Protein Science. 2014 Oct;23(10): 1469-896X. pp. 1341 – 1348. 2014. doi: 10.1002/pro.2518. PMID: 25043951. PMCID: PMC4286996.
Islet Amyloid Polypeptide (IAPP) is a 37-residue hormone cosecreted with insulin by the β-cells of the pancreas. Amyloid fiber aggregation of IAPP has been correlated with the dysfunction and death of these cells in type II diabetics. The likely mechanisms by which IAPP gains toxic function include energy independent cell membrane penetration and induction of membrane depolarization. Although the relationship between amyloid formation and toxicity is poorly understood, the fact that conditions promoting one also favor the other suggests related membrane active structural states. This paper describes a novel high throughput screening protocol that capitalizes on this correlation to identify compounds that target membrane active species. Out of 960 known bioactive compounds screened, 37 compounds were identified as active toward IAPP fiber formation. Several compounds tested in secondary cell viability assays also demonstrate cytoprotective effects. YCMD contributed to the publication by assisting in designing and performing the screen of the MicroSource Gen-Plus library described in the publication.
|
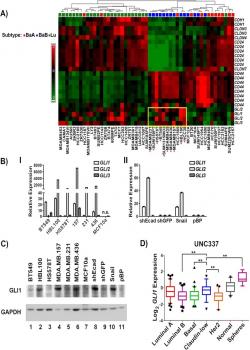 |
Colavito S, Zou MR, Yan Q, Nguyen DX, Stern DF. Significance of glioma-associated oncogene homolog 1 (GLI1) expression in claudin-low breast cancer and crosstalk with the nuclear factor kappa-light-chain-enhancer of activated B cells (NFκB) pathway. Breast cancer research 2014 Sep 25;16(5):444. doi: 10.1186/s13058-014-0444-4 : PMID: 25252859. PMCID: PMC4303124.
The recently identified claudin-low subtype of breast cancer is enriched for cells with stem-like and mesenchymal-like characteristics, has a poor prognosis, and has few targeted treatment options. Using a high throughput inhibitor screen at YCMD, we identified high expression of glioma-associated oncogene homolog 1 (GLI1), the effector molecule of the hedgehog (Hh) pathway, as a critical determinant of cell lines that have undergone an epithelial to mesenchymal transition (EMT). Our results uncover crosstalk between NFκB and GLI1 signals and suggest that targeting these pathways may be effective against the claudin-low breast cancer subtype.
|
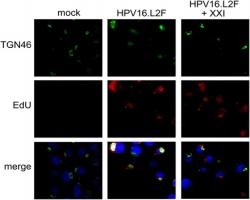 |
Zhang W, Kazakov T, Popa A, Goodwin EC, DiMaio D. Vesicular trafficking of incoming human papillomavirus 16 to the Golgi apparatus and endoplasmic reticulum requires g-secretase activity. MBio 2014 Sep 16; 5(5):e01777-14. doi:10.1128/mBio.01777-14. 2014. PMID:25227470. PMCID: PMC4172078.
This paper presents follow up studies performed on hits identified in an siRNA screen conducted at YCMD.
|
 |
Pirruccello M, Nandez R, Idevall-Hagren O, Alcazar-Roman A, Abriola L, Berwick SA, Lucast L, Morel D, DeCamilli P Identification of inhibitors of inositool 5-phosphatases through multiple screening strategies. ACS Chem Biol. 2014 Jun 20;9(6):1359-68. doi: 10.1021/cb500161z. PMID: 24742366. PMCID: PMC4076014.
Small molecules that inhibit or activate these enzymes have potential therapeutic applications and will be powerful research tools. This paper describes a high throughput biochemical screen performed at YCMD and the follow up assays that were done in the hits. Work performed at YCMD included developing three complimentary in vitro assays against multiple enzymes and substrates, high throughput screening against two enzymes, hit triaging/clustering/expansion and testing hit compounds against a panel of 5-phosphatases and various substrates. Two chemical scaffolds were identified and one was active in cells.
|
 |
YCMD has conducted a comprehensive meta-analysis of FDA approved drugs published in a series of Drug Discovery Today manuscripts.
Click here to see the complete listing of publications.
|
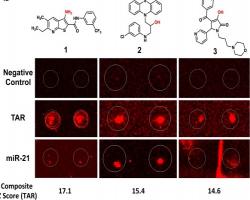 |
Sztuba-Solinska J, Shenoy SR, Gareiss P, Krumpe LRH, Le, Grice SFJ, O’Keefe BR, Schneekloth Jr. JS. Identification of Biologically Active, HIV TAR RNA-Binding Small Molecules Using Small Molecule Microarrays. J Am Chem Soc. 2014 May 12; 136: 8402-8410. doi:10.1021/ja502754f. PMID: 24820959. PMCID: PMC4227816.
Identification of RNA binding small molecules remains an important therapeutic challenge. YCMD employed small molecule microarray technology to screen for compounds targeting the transactivation response RNA hairpin of HIV. A compound was identified that binds to this structured RNA target, and follow-up studies with scientists from the NCI showed that this compound is able to inhibit HIV induced cellular toxicity.
|
 |
Mannam P, Shinn AS, Srivastava A, Neamu RF, Walker WE, Bohanon M, Merkel J, Kang MJ, Dela Cruz CS, Ahasic AM, Pisani MA, Trentalange M, West AP, Shadel GS, Shen J, Elias JA, Lee PJ. MKK3 regulates mitochondrial biogenesis and mitophagy in sepsis-induced lung injury. Am J Physiol Lung Cell Mol Physiol. 2014 Apr 1; 306(7):L604-19. doi: 10.1152/ajplung.00272.2013. PMID: 24487387. PMCID: PMC3962628.
Peripheral blood mononuclear cells were assayed at YCMD for activation of MKK3 (using a phospho-MKK3/6 SureFire assay), and compared between critically ill septic patients and non-septic patients. MKK3 is a kinase involved in sepsis and mitochondrial function. MKK3 deficiency is protective and reduces the generation of reactive oxygen species.
*Chosen Best Research Paper Published in AJP-Lung by a Junior Investigator
|
 |
Albright RA, Ornstein DL, Cao W, Chang WC, Robert D, Tehan M, Hoyer D, Liu L, Stabach P, Yang G, De La Cruz EM, Braddock DT. Molecular basis of purinergic signal metabolism by ectonucleotide pyrophosphatase/phosphodiesterases 4 and 1 and implications in stroke. J Biol Chem. 2014 Feb 7;289(6):3294-306; doi: 10.1074/jbc.M113.505867. PMID: 24338010. PMCID: PMC3916532. |
|

|
Neklesa TK, Noblin DJ, Kuzin A, Lew S, Seetharaman J, Acton TB, Kornhaber G, Xiao R, Montelione GT, Tong L, Crews CM. A bidirectional system for the dynamic small molecule control of intracellular fusion proteins. ACS Chem Biol. 2013 Oct 18;8(10):2293-300. doi: 10.1021/cb400569k. PMID: 23978063. PMCID: PMC4113957.
Small molecule control of intracellular protein levels allows temporal and dose-dependent regulation of protein function. The Crews laboratory screened the YCMD compound library and identified compounds that can both degrade or stabilize fusion proteins of interest.
|
Sayegh J, Cao J, Zou MR, Morales A, Blair LP, Norcia M, Hoyer D, Tackett AJ, Merkel JS, Yan Q. Identification of Small Molecule Inhibitors of Jumonji AT-Rich Interactive Domain 1B (JARID1B) Histone Demethylase by a Sensitive High Throughput Screen. J Biol Chem. 2013; 288: 9408-9417. doi: 10.1074/jbc.M112.419861. PMID: 23408432. PMCID: PMC3611010.
JARID1B (also known as KDM5B or PLU1) is a member of the JARID1 family of histone lysine demethylases responsible for the demethylation of trimethylated lysine 27 in histone H3 (H3K4me3), a mark for actively transcribed genes. JARID1B is an attractive target for cancer therapy. Working with YCMD, the Yan lab developed and optimized a high-throughput ALPHAscreen assay for KDM5B activity. Over 15,000 small molecules were screened and several novel KDM5B inhibitors were identified with low micromolar inhibitory activity, in addition to several known demethylase inhibitors. These new KDM5B inhibitors are being further investigated and optimized.
|
|
 |
Grotzke JE, Lu Q, Cresswell P. Deglycosylation-dependent fluorescent proteins provide unique tools for the study of ER-associated degradation. Proc Natl Acad Sci USA. 2013 Feb 26;110(9):3393-8. PMID: 23401531. PMCID: PMC3587246.
Endoplasmic reticulum–associated degradation (ERAD) is a constitutive process that identifies misfolded proteins in the ER and shuttles them to the cytosol, where they can be degraded by the proteasome. This paper reports a novel system to analyze ERAD based on mutants of split or intact Venus fluorescent protein for which fluorescence depends on enzymatic deglycosylation. Genomewide siRNA screen was performed at YCMD and successfully identified known ERAD factors, validating performance of the system and showing that it could greatly facilitate the identification of ERAD components and investigation of this critical pathway.
|
 |
Held, Matthew A, Langdon, Casey G, Platt, James T, Graham-Steed, Tisheeka, Liu, Zongzhi, Chakraborty, Ashok, Bachinocchi, Antonella, Koo, Andrew, Haskings, Jonathan W, Bosenberg, Marcus W, Stern, David F. Genotype-Selective Combination Therapies for Melanoma Identified by High-Throughput Drug Screening. Cancer Discovery. 2013 January 3;1 1-3; doi:10.1158/2159-8290.CD-ITI13-01. PMID: 23239741. PMCID: PMC3546137.
Single agent dose response testing was performed at YCMD to identify three concentrations to be used in pairwise drug testing against a large panel of patient-derived melanoma cell lines. The combination drug plates were made at YCMD and used in the screening. Many important insights regarding molecular phenotype and drug responsiveness were observed.
|
 |
Lipovsky A, Popa A, Pimienta G, Wyler M, Bhan A, Kuruvilla L, Guie MA, Poffenberger AC, Nelson CD, Atwood WJ, DiMaio D. Genome-wide siRNA screen identifies the retromer as a cellular entry factor for human papillomavirus. Proc Natl Acad Sci USA. 2013 Apr 30;110, 7452-7. PMID: 23569269. PMCID: PMC3645514.
This paper describes a genome-wide siRNA screen which was performed to identify cellular genes required for human papillomavirus (HPV) biology. The screen identified numerous potential antiviral targets and many retrograde transport factors, including multiple subunits of the retromer. High-throughput image-based siRNA assay development, whole-genome screening and screen follow-up were conducted at YCMD.
|
 |
Albright, RA, Chang, WC, Robert D, Ornstein DL, Cao W, Redick ME, Young JI, De La Cruz EM, Braddock DT, 2012. NPP4 is a procoagulant enzyme on the surface of vascular endothelium. Blood. 2012 Nov 22; (22):4432-40.doi: 10.1182/blood-2012-04-425215. PMID: 22995898. PMCID: PMC4017314.
|
|

|
Sayegh J, Cao J, Zou MR, Morales A, Blair LP, Norcia M, Tachkett AJ, Merkel JS, Yan Q. Identification of small molecule inhibitors of Jumonji AT-rich interactive domain 1B (JARID1B) histone demethylase by a sensitive high throughput screen. J Biol Chem. 2013 Mar 29;288(13):9408-17. doi: 10.1074/jbc.M112.419861. PMID: PMC3611010.
A new assay was developed at YCMD and used to screen 15,000 small molecules for targets overexpressed in cancers. The chemiluminescent assay helped identify inhibitors for this important and emerging class of epigenetic targets.
|
 |
Bannwarth L, Goldberg AB, Chen C, Turk BE. Identification of exosite-targeting inhibitors of anthrax lethal factor by high-throughput screening. Chem Biol, 2012 Jul 27;19(7):875-82. doi: 10.1016/j.chembiol.2012.05.013. PMID: 22840775. PMCID: PMC3408624.
Development and screening of fluorescence polarization assay was conducted at YCMD. A Center chemist synthesized modified depsidones for Structure Activity Relationship studies that inhibit protease activity.
|
 |
Buckley DL, Gustafson JL, Van Molle I, Roth AG, Tae HS, Gareiss PC, Jorgensen WL, Ciuli A, Crews CM. Small-molecule inhibitors of the interaction between the E3 ligase VHL and HIF1α. Angew Chem Int Ed Engl. 2012 Nov 12;51(46):11463-7. doi: 10.1002/anie.201206231. PMID: 23065727. PMCID: PMC3519281.
Protein-protein interactions (PPIs) are vital to most biological processes, yet despite recent advances they remain notoriously difficult to target. The biologically relevant interaction between the E3 ubiquitin ligase VHL and HIF1alpha regulates hypoxia driven gene expression. Utilizing a fluorescent polarization based assay at YCMD, SAR studies of small molecule modulators of this interaction were investigated. In addition, computational chemistry methods were utilized to guide the design of new small molecule inhibitors of this interaction.
|
 |
Noblin, DJ, Page, CM, Tae, HS, Gareiss, PC, Schneekloth, JS, Crews, CM. A HaloTag-Based Small Molecule Microarray Screening Methodology with Increased Sensitivity and Multiplex Capabilities. ACS Chemical Biology, 2012 Dec 21; 7(12) 2055-2063, doi: 10.1021/cb300453k. PMID: 23013033. PMCID: PMC3547077.
Small molecule microarray screening provides an affinity based method to determine compound interaction with targets of interest. Traditionally, targets of interest are detected using antibodies or direct fluorescent labels. In collaboration with YCMD, a new method of target identification utilizing the HaloTag technology was shown to have increased sensitivity in addition to multiplex capabilities.
|
 |
Raycroft MT, Harvey BP, Bruck MJ, and Mamula MJ. Inhibition of Antigen Trafficking through Scavenger Receptor A, The Journal of Biological Chemistry. 2012 Feb 17;287(8):5310-6. doi: 10.1074/jbc.M111.316356. PMID: 22215667. PMCID: PMC3285311.
Two small molecule inhibitors that reduce T cell immunity and antigen transfer were identified from an assay developed and screened at YCMD. Molecules such as these may help halt autoimmune disease development.
|
 |
Woo CM, Beizer NE, Janso JE, Herzon SB. Isolation of Lomaiviticins C−E, Transformation of Lomaiviticin C to Lomaiviticin A, Complete Structure Elucidation of Lomaiviticin A, and Structure−Activity Analyses. J Am Chem Soc. 2012 Sep 19;134(37), 15285-8. PMID: 22963534.
|
 |
Freed EF, Prieto JL, McCann KL, McStay B, Baserga SJ. NOL11, implicated in the pathogenesis of North American Indian childhood cirrhosis, is required for pre-rRNA transcription and processing. PLoS Genet. 2012 Aug 8(8) doi: 10.1371/journal.pgen.1002892. PMID: 22916032. PMCID: PMC3420923.
This paper describes yeast two-hybrid screen identifying a novel nucleolar protein NOL11, and presents a variety of assays showing that NOL11 is required for the early stages of ribosome biogenesis in humans. siRNA NOL11 knockdown followed by nucleolar staining and imaging assays were conducted at YCMD and confirmed the role of NOL11 in ribosome biogenesis and determining nucleolar morphology.
|
 |
Cho Y, Vermeire JJ Merkel JS, Leng L, Du X, Bucala R, Cappello M, Lolis, E. 2012. Drug repositioning and pharmacophore identification in the discovery of hookworm MIF inhibitors, Chemistry & Biology. 2011 Sep 23; 18(9) 1089-1101. PMID: 21944748; PMCID: PMC3294498.
Assays were developed at YCMD using recombinant MIF from hookworm parasites were screened for enzymatic activity against bioactive libraries. Hookworm-specific inhibitors were identified. Investigators solved co-crystal structures of inhibitor and enzyme. |
 |
Braddock, D.T. Autotaxin and lipid signaling pathways as anticancer targets. Current Opinion in Investigational Drugs. 2010; 11: 629-37. PMID: 20496257.
|
 |
Gareiss PC, Schneekloth AR, Salcius MJ, Seo S-Y, Crews CM. Identification and Characterization of a Peptidic Ligand for Ras. ChemBioChem 2010; 11:517-22. PMID: 20082398
Ras is oncogenic GTPase that remains an elusive drug target. Utilizing phage display, a small peptidic ligand for Ras was identified. A fluorescence polarization assay was developed with YCMD and used to characterize the molecular interaction. Additionally an enzymatic assay was developed to determine the effects of the peptide on the GTPase function of Ras.
|
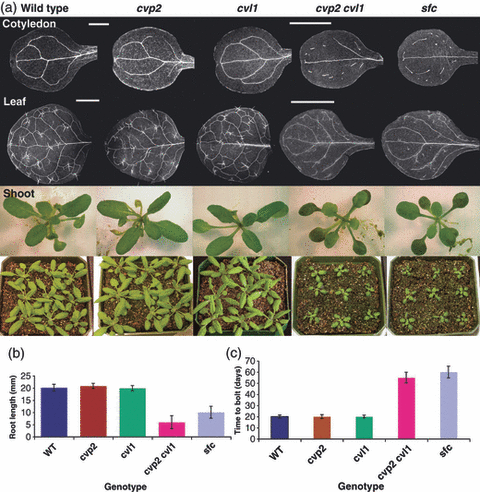 |
Carland F, Nelson T. CVP2- and CVL1-mediated phosphoinositide signaling as a regulator of the ARF GAP SFC/VAN3 in establishment of foliar vein patterns. Plant J. 2009 Sep;59(6):895-907. doi: 10.1111/j.1365-313X.2009.03920.x. PMID:19473324. |
 |
Goodwin, EC, Atwood, WJ, and DiMaio, D. High-throughput, cell-based screen for chemicals that inhibit infection by SV40 and human polyomaviruses. J. Virol 2009; Jun; 83(11)5630-5639. PMID: 19297472; PMCID: PMC2681952.
|
 |
Yi F, Zhu P, Southall N, Inglese J, Austin CP, Zheng W, Regan L. An AlphaScreen-Based High-Throughput Screen to Identify Inhibitors of Hsp90-Cochaperone Interaction. J Biomol Screen. 2009 Mar 14; 14(3) 273-81. PMID: 19211782; PMCID: PMC3066041.
|
 |
Rodriguez-Gonzalez A, Cyrus K, Salcius M, Kim K, Crews CM, Deshaies RJ, Sakamoto KM. Targeting steroid hormone receptors for ubiquitination and degradation in breast and prostate cancer. Oncogene. 2008 Dec 4;27(57):7201-11. PMID: 18794799. PMCID: PMC5573236.
|
 |
Lewis CA, Merkel J, Miller SJ. Catalytic site-selective synthesis and evaluation of a series of erythromycin analogs. Bioorg Med Chem Lett. 2008 Nov 15; 18(22) 60007-6011. PMID: 11819795. PMCID: PMC2669672.
|
 |
Saunders LP, Ouellette A, Bandle R, Chang WC, Zhou H, Misra RN, De La Cruz EM, Braddock DT. Identification of small-molecule inhibitors of autotaxin that inhibit melanoma cell migration and invasion. Mol Cancer Ther. 2008 Oct; 7(10):3352-3362. PMID: 18852138.
|
 |
Blount K, Puskarz I, Penchovsky R, Breaker R. Development and application of a high-throughput assay for glmS riboswitch activators. RNA Biol. 2006 Apr;3(2):77-81. PMID: 17114942.
|